19) Select the major product of the following series of reactions. MgBr 1) CO₂(s) 2) H₂O* 3) CH₂CH₂OH, H', heat ? 20) Select the major product of the following reaction. де 1) LiAlH4 2) H₂O OH a) H₂N OH ? 21) Select the major product of the following series of reactions. OH 1) SOCI₂, heat 2) (CH3CH2)2CuLi 3) H₂O ? OH d) OH b) d) HO ol of de o
19) Select the major product of the following series of reactions. MgBr 1) CO₂(s) 2) H₂O* 3) CH₂CH₂OH, H', heat ? 20) Select the major product of the following reaction. де 1) LiAlH4 2) H₂O OH a) H₂N OH ? 21) Select the major product of the following series of reactions. OH 1) SOCI₂, heat 2) (CH3CH2)2CuLi 3) H₂O ? OH d) OH b) d) HO ol of de o
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter10: Alcohols
Section: Chapter Questions
Problem 10.50P
Related questions
Question
Do question 19, and or any other questions on the page. Please explain
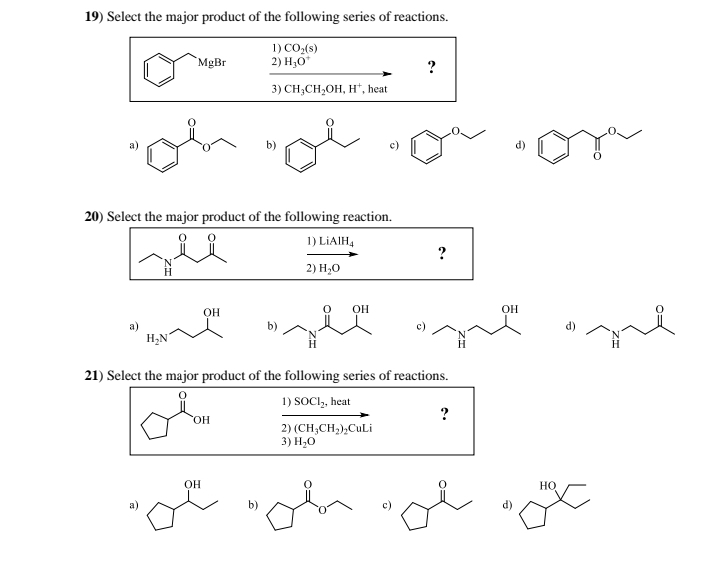
Transcribed Image Text:19) Select the major product of the following series of reactions.
MgBr
1) CO₂(s)
2) H₂O*
3) CH₂CH₂OH, H', heat
?
20) Select the major product of the following reaction.
де
1) LiAlH4
2) H₂O
OH
a)
H₂N
OH
?
21) Select the major product of the following series of reactions.
OH
1) SOCI₂, heat
2) (CH3CH2)2CuLi
3) H₂O
?
OH
d)
OH
b)
d)
HO
ol of de o
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 1 steps

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning