Chapter1: Numerals And Fractions
Section: Chapter Questions
Problem 5RP
Related questions
Question
For the following reactions, identify the atom(s) being oxidized and reduced:
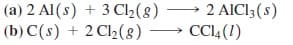
Transcribed Image Text:2 AICI3(s)
(a) 2 Al (s) + 3 Cl2(8)
(b) C(s) + 2 Cl½(8)
Expert Solution
Step 1
A redox reaction is one in which both oxidation and reduction take place simultaneously. When a substance gains oxygen or loses electrons, it is said to have undergone the process of oxidation and contrarily when a substance loses oxygen or gains electrons, it is said to have undergone the process of reduction. In a redox reaction, one compound is oxidized while the other is reduced. Oxidation number is the number of electrons an atom has gained or lost in comparison to a neutral atom. Calculation of the oxidation number would be helpful in determining is a compound has undergone oxidation or reduction during a chemical reaction.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, nursing and related others by exploring similar questions and additional content below.Recommended textbooks for you



Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning



Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College