Q: Test for Ethers a. Ferrox paper test: General Reaction Reagents: Positive result: Notes: Test for…
A: Ferrox test used for identify the presence of oxygen. Hence, this test is used for oxygen containing…
Q: CEN + ? MgBr (1). LIAIH MgBr b). MgBr c). ) d). LIAKO-t-Bu)3 e) DIBAH
A:
Q: Give the structures of the aldol products that form when each of the following compounds or mixtures…
A: 1. Given that, Butanal, Cyclopentanone, and Acetophenone. We have to draw the self aldol product of…
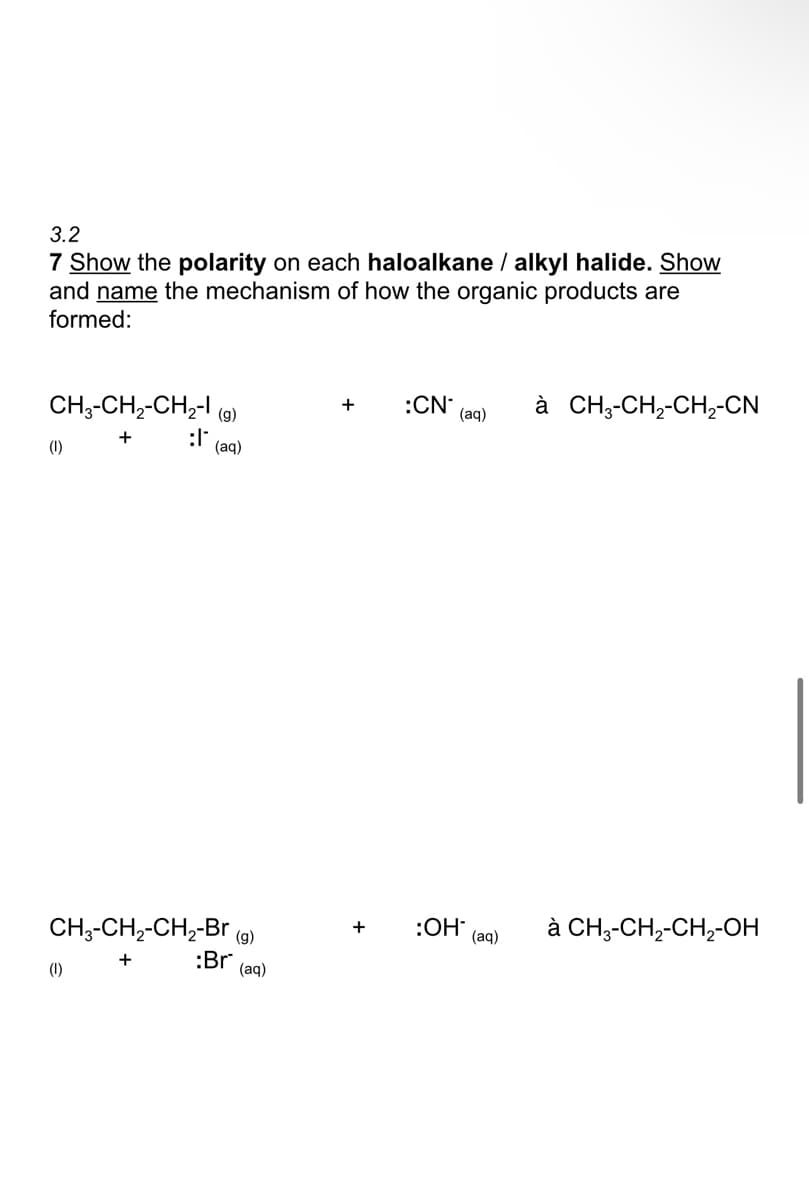
Q: cyclohery! ethyl ether CH3 CH2 Br , Br CH, CH,O°
A: There are generally two mechanism for nucleophilic substitution reaction- 1- SN1 reaction 2- SN2…
Q: 4. Name the chemicai test and state the expected resuit that couid be used to distinguish the…
A:
Q: complete electronic configuration of Ge.
A:
Q: The decomposition of N,O5 to NO, and O, is first order, with a rate constant of 4.80 x 10-4/s at 45°…
A: Answer: a) 1.11×10^-2 mol/L, b) 1043.5 s, last part) 1064.4 s
Q: Following ester (methyl benzoate) was hydrolyzed in presence of an acid catalyst. This reaction…
A:
Q: Part A Consider the following reaction: 2 Co(g) + 2 NO(g) - 2 CO2(g) + N2(g) Calculte the standard…
A: Calculate the standard enthpy change of the given reaction by using table given above--->
Q: Name this compound: HOCCHCH, ÓCH3
A: Note : Since you have posted multiple questions, we are entitled to answer the first only. Please…
Q: Name the Following Compounds
A:
Q: Diethylamine (structure presented below) and HCl react to produce CH3 CH3 diethylammonium chloride…
A:
Q: Molecule Name (or short form name) of molecule Order the molecule is in during Glycolysis compared…
A:
Q: Question 1 Identify the correct statements consistent with the following scenario: 1. Heat is lost…
A: The correct statements have been marked in the following step.
Q: The maximum amount of calcium carbonate that will dissolve in a 0.243 M calcium acetate solution is…
A:
Q: Which element has an quantum angular moment (L) of 0. OU O Mg O Fe
A: there are 4 types of quantum numbers- 1- principal quantum number 2- angular moment quantum number…
Q: Calculate the concentration of all the ions in solution after mixing 100.0 mL of 0.0200M KF (aq)…
A:
Q: Which of these alkenes show cis-trans isomerism? For each that does, draw structural formulas for…
A: The alkene will show cis and transfer if it has two condition- 1- Different group should be present…
Q: Fill in the nuclide symbol for the missing particle in the following nuclear equation. 218 At -je+At
A: Given :- ? → -1eo + 85At218 To determine :- nuclide symbol of missing species
Q: Dinitrogen pentoxide decomposes acconting to the reaction below. What is the rate of appearance O,…
A: The answer is given below
Q: What is the binding energy in kJ/mol nucleons for lanthanum-138? |k]/mol nucleons 57 H+ 81;n La The…
A: The reaction given is, =>
Q: Some Radioactive Isotopes Useful in Medical Imaging Mode of Decay Use in Medical Imaging Isotope…
A: (1) We have to tell the number of protons and the number of neutrons present on the radioactive…
Q: A vessel containing 3 mol of an ideal gas, initially at 4 atm and 50 °C, is heated until the…
A: The answer is given below
Q: 7.00 grams of Cobalt (II) nitrate are dissolved in 1.00 liter of water. What is the initial…
A:
Q: (i) Give the molecular, structural and displayed formulae of the following molecules: (a) 2, 2 –…
A:
Q: Check the box before each formula that represents a ligand that is likely to form a complex with a…
A: Ligands are the species which donates lone pair to central metal atom or ion.
Q: = F = HF
A: Weak acids are those acids which do not ionize completely to give H+ ions (that is the extent of…
Q: A student is given a 0.1 M solution of an acid and is asked to determine whether it is a solution of…
A: Given: Concentration of acid = 0.1 M And the pH of the solution = 2.3
Q: Which of the following describe(s) the following reaction? Choose all that apply. 14N + He 70 + H O…
A: The reaction given is,
Q: nductemearterie delasgtsecunca e reacoone CH,CI Br HNO, AICI, FeBr, H,SO, NO ON NO NO, 2.
A:
Q: Which of the following describes the reaction: 218 Po →°,e+ 218 At 84 ³Po→,e+ 85
A: In above reaction atomic number on product side increase by one while mass number remains unchanged.…
Q: t Was the IIItial tenperature in C? tem has initial volume and temperature of 8160mL and 467.0K. !f…
A: Given-> V1 = 8160 ml T1 = 467.0 K T2 = -151°C = -151 + 273 = 122K
Q: Find the molarity of a KOH
A:
Q: A certain third order reaction has a rate of 0.11 M/s when the concentration of reactant A is 0.15…
A:
Q: 1.What is the advantage of using 12 in this experiment? 2. Why is the concentration of lodine much…
A: The iodination of acetone can be represented as:
Q: Br. FeBr, CH Br 2.
A:
Q: Which of the following describes the reaction: ° Bi → He + 0 TI 81 83 alpha decay reaction O beta…
A:
Q: Prove: (a) Hermitian matrices have real eigenvalues only. (b) (Normalized) eigenvectors of hermitian…
A:
Q: Iodine-131 captures an electron and emits 3 positrons in one of its decay processes. What is the…
A:
Q: For which of the mixtures will Ag2SO4(s) precipitate? 150.0 mL of 0.10 M Na2SO4(aq) and 5.0 mL of…
A: To identify: The mixture for which Ag2SO4 will precipitate.
Q: A sample of helium gas has a volume of 6.30 L at a pressure of 825 mmHg and a temperature of 21∘C.…
A: Given: The initial volume, pressure, and temperature of helium gas are 6.30 L, 825 mmHg, and 21 ∘C…
Q: Above what Fe2+ concentration will Fe(OH)2 precipitate from a buffer solution that has a pH of 8.10?…
A: Answer: 3.07×10^-5 M
Q: Aldol Product FOM. OMe 7.38 ppm pom 7.9 7o 72 3.91 ppm Ppm 4. 3.9 ppm 10
A: The proton NMR helps us to find out the different types of proton present in an unknown organic…
Q: Draw the major organic product(s) of the following reaction. CI CH3OH NaOCH,
A:
Q: 5. Differentiate one from the other.
A: The first question (Q4) has already been answered. As only one question can be answered as per the…
Q: Silver nitrate + ammonium hydroxide
A: Reaction between Silver nitrate + ammonium hydroxide -----
Q: 21.) Calculate the enthalpy of hydrogenation of benzene to cyclohexane from the following reactions…
A: The reactions given are, 1) C6H6 (l) + 15/2 O2 (g) → 6 CO2 (g) + 3 H2O (l) ΔH…
Q: Complete the following nuclear bombardment equation by filling in the nuclear symbol for the missing…
A: Complete the following nuclear reaction-->
Q: Example 8: An aqueous solution is 0.907 M Pb(NO3),. Determine the molality of lead (II) nitrate,…
A:
Q: At STP conditions, 25 mL of hydroiodic acid generates 1.20 L of H2 gas when combined with excess…
A: Given-> Volume of HI = 25 ml = 0.025 L (1 L = 1000 ml) Volume of H2 gas = 1.20 L At STP->…
Step by step
Solved in 2 steps with 1 images

- Oximene and myrcene, two hydrocarbons isolated from alfalfa that have the molecular formula C10H16, both yield 2,6- dimethyloctane when treated with H2 and a Pd catalyst. Ozonolysis of oximene forms (CH3)2C = O, CH2 = O, CH2(CHO)2, and CH3COCHO. Ozonolysis of myrcene yields (CH3)2C = O, CH2 = O, (two equiv), and HCOCH2CH2COCHO. Identify the structures of oximene and myrcene.The crocodile, which can remain under water without breathing for up to 1 h, drowns its air-breathing prey and then dines at its leisure. An adaptation that aids the crocodile in doing so it that it can utilize virtually 100% of the O2in its blood whereas humans, for example, can extract only ~65% of the O2in their blood. Crocodile Hb does not bind BPG. However, crocodile deoxyHb preferentially binds HCO3ꟷ. How does this help crocodile obtain its dinner? Explain your answer.Benzene is one of the compounds used as octane enhancers in unleaded gasoline. It is manufactured by thecatalytic conversion of acetylene to benzene: 3C2 H2(g) ⇌ C6 H6(g). Which value of Kc would make this reactionmost useful commercially? Kc ≈ 0.01, Kc ≈ 1, or Kc ≈ 10. Explain your answer
- The above reaction involves heterolytic bond breakage of HBr a) Encircle the nucleophile (s) and electrophile (s) and explain why. b) Give the mechanism of the reaction by: ) Drawing the appropriate arrows to track the flow of electrons in Showing the species formed after bond breakage and bond formation with appropriate charges. c) Predict the final products.Based on the more stable conformation (conformation A), need help providing two mechanisms for the E2 elimination of the protonated version of cis-1 using a general base (B:) to give alkenes 2 and 3. Clearly show which hydrogens meet the stereochemical requirements for E2-elimination Using Zaitsev’s rule, indicate which is favoured. Pictured is also a reaction mechanism to show what the alkenes 2 and 3 are. Thank you :)What organic product is formed when 1‑methylcyclopentene is treated with NMMO in the presence of H2O and a catalytic amound of OsO4? Clearly show stereochemistry by drawing a wedge and dashed bond for each chiral carbon. Only draw one stereoisomer if more than one can be formed.
- Draw all of the substitution and elimination products formed from thegiven alkyl halide with each reagent: (a) CH3OH; (b) KOH. Indicate thestereochemistry around the stereogenic centers present in the products,as well as the mechanism by which each product is formed.2-bromopentane, when treated with alcoholic KOH yields a mixture of three alkenes A, B and C. Identify A, B and C. Which is predominant?Draw the organic products formed when cyclopentene is treated with each reagent. With some reagents, no reactionoccurs.a. H2 + Pd-Cb. H2 + Lindlar catalystc. Na, NH3d. CH3CO3He. [1] CH3CO3H; [2] H2O, HO–f. [1] OsO4 + NMO; [2] NaHSO3, H2Og. KMnO4, H2O, HO–h. [1] LiAlH4; [2] H2Oi. [1] O3; [2] CH3SCH3j. (CH3)3COOH, Ti[OCH(CH3)2]4, (–)-DETk. mCPBAl. Product in (k); then [1] LiAlH4; [2] H2O
- An optically active alkyne A (C10H14) can be catalytically hydrogenated to butylcyclohexane. Treatment of A with EtMgBr liberates no gas. Catalytic hydrogenation of A over Pd/C in the presence of quinoline poison and treatment of the product with O3 and then H2O2 gives an optically active tricarboxylic acid C8H12O6 (A tricarboxylic acids is a compound with three - CO2H groups.). Give the structure of A, and account for all observations.Reaction of 2-methyl-2-pentene with reagent is regioselective. Draw a structural formula for the product of reaction and account for the observed regioselectivity. Q.) Br2 in H2Oγ-Butyrolactone (C4H6O2, GBL) is a biologically inactive compound that is converted to the biologically active recreational drug GHB (Section 19.5) by a lactonase enzyme in the body. Since γ-butyrolactone is more fat soluble than GHB, it is more readily absorbed by tissues and thus produces a faster onset of physiological symptoms. γ-Butyrolactone shows an absorption in its IR spectrum at 1770 cm−1 and the following 1H NMR spectral data: 2.28 (multiplet, 2 H), 2.48 (triplet, 2 H), and 4.35 (triplet, 2 H) ppm. What is the structure of γ-butyrolactone?

