Q: Calculate the standard free energy, ΔGo, for the following equation at 25.0 oC and determine if the…
A: DGf° (C2H6(g)) + 7Cl2(g) ⟶ 2CCl4(g) + 6HCl(g) DGf° (C2H6(g)) = -32.82 kJ/mol DGf° (Cl2(g)) = 0…
Q: Fasagraph Using the following data (at 25 °C): Camond (s) +02(g) Craphitel S) O2(g) Cyraphite 6.…
A: As per given question, we need to find the heat of formation of graphite For the formation of…
Q: 2NO(g)+O2(g)→2NO2(g)ΔH°=−112kJ/molrxn Answer the following questions about the reaction represented…
A: Please have a look on the sol:
Q: is the free energy change for the reaction below, if the reaction quotient, Q, at 25.0oC is equal to…
A: Given-> Reaction quotient (Q) = 4.56 × 10-6 ∆G° = -216.790 KJ = -216.790 × 1000 J = -216790 J (1…
Q: Predict the sign of ΔS° for(a) 2K(s) + F2(g) → 2KF(s)(b) NH3(g) + HBr(g) → NH4Br(s)(c) NaClO3(s) →…
A: The sign of the Standard change in entropy is obtained from the difference in the moles of product…
Q: Given the following enthalpy change values for the reactions below: A + B -->2C; ?rH = 419 kJ mol-1…
A: According to the hess's law we know that when a chemical equation is written as the sum of overall…
Q: Predict the signs of ΔH and ΔS for this reaction. Explainyour choice.
A: Given diagram,
Q: A student determines the value of the equilibrium constant to be 7.86x10-37 for the following…
A: Given-> Keq = 7.86 × 10-37 Moles of H2S = 2.28 moles
Q: Calculate ∆ Grxn° values for the following reactions at 298 K using the tabulated ∆ Gf° values. a)…
A: The chemical reactions given are a) 2 Mg (s) + O2 (g) → 2MgO(s) −1138.6 kJ/mol b) 2 CH3OH (g) + 3…
Q: What is K for a reaction if ∆G° =-369.4 kJ/mol at 25.0 °C? (R = 8.314 J/mol ・ K)
A: 1- First converted ∆G° unit kJmol-1 to Jmol-1 ∆G° = -369. 4 kJmol-1 We know that, 1kJ = 1000 J…
Q: 3. Calculate the standard free energy of formation, AGº, at 50°C, for each of the following…
A: a)
Q: Calculate the ∆?° at 298K of the following reaction, H2(g) + F2(g) ----> 2HF(g). Given that our ∆H°…
A:
Q: 5. Calculate AS° values for the following reactions by using tabulated S° values from Appendix C. In…
A: 5. (a) HNO3(g) + NH3(g) ---> NH4NO3(s) The entropy value S° given are - S°(HNO3) = 266.4 J/K…
Q: Calculate AH for below reaction at 25C C(s) + 2H2(g) ==> CH4 (g) if AU =511.24 Cal, R = 1.987…
A:
Q: (22) Using the provided tabulated values, calculate AS"ran for the following reaction that occurs at…
A: Options : C) 278.2 k/J Given : Following Reaction, CS2(g) + 3Cl2(g) → CCl4(g) + S2Cl2(g)…
Q: For the following relationship: a) determine the KC value of that reaction by reaching equilibrium…
A: The enthalpy change of the given reaction at 25°C is -18 kcal/mol and the entropy change of the…
Q: What is the sign of delta S for each Enter anser as + or - A) the sublimation of 1 mol H2O(s) to…
A: Since we know that ∆S depends upon on following factors (1) When the phase of a substance change…
Q: Indicate wheather the reation is thermodynamically favored at 25°C. Justify your answer.
A: Answer
Q: 3c. Calculate AH for the following reaction, FeO(s) + CO(g) Fe(s) + CO2(g), from the following data,…
A:
Q: AS is negative for which of the following reactions? H2O (1) --> H2O (g) O2C (s) +O2 (g) --> 2 CO…
A:
Q: Comment on whether ΔH and ΔS favor spontaneity of the following reactions. Explain your reasoning!…
A: Spontaneous reaction are those which occur in forward direction and favour the formation of product.
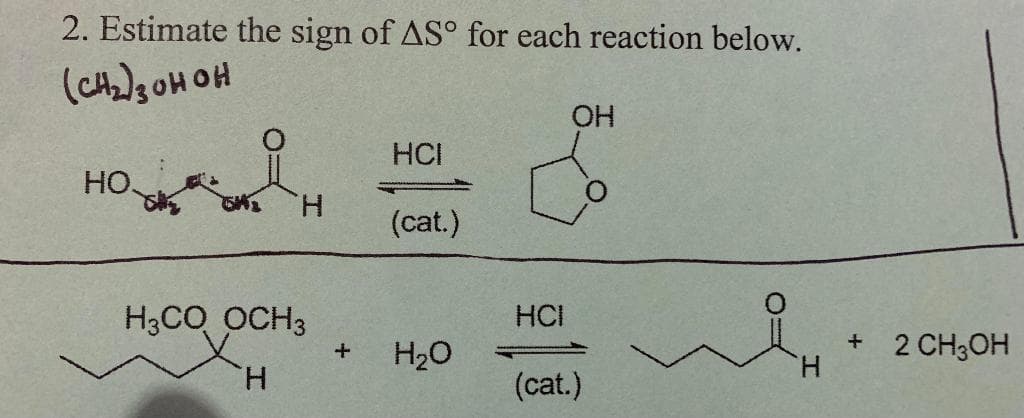
Q: CH3 -OCH3 + Br С. + Br Predict the sign for C. AS° is negative AS° is & 0 AS° is positive
A: Here no of reactant and product both is 2 ∆S0=No of product -No of reactant
Q: A free energy diagram for a generic reaction, reactants products, shown below. On the horizontal…
A: We are given a graph of Gibbs free energy of reaction(Grxn) versus the extent of reaction.
Q: Determine the value of AG° at 25 °C for the reaction below given the following data: CH,(g) AG = 68…
A:
Q: Calculate ΔS° for the reaction of H2(g) with liquid benzene (C6H6) to give cyclohexane (C6H12). ?°…
A:
Q: Part B Give AH for reaction above. Express your answer as an integer. ? kJ
A: b. Given reaction is: C3H8(g)+5O2(g)→3CO2(g)+4H2O(g)+2044 kJ From the reaction, The heat can be seen…
Q: Given the following at 25 "C and 1.00 atm: AHP = 33.2 kJ AHP = 11.1 kJ 1/2N2(g) + O2(g) - NO2(g)…
A: Given equations are : 1/2 N2 (g) + O2 (g) -------> NO2 (g) , ΔH1 = 33.2 KJ N2 (g) + 2O2 (g)…
Q: What is K for a reaction if ∆G° =-237.2 kJ/mol at 25°C (or 298 K)? (R = 8.314 J/mol ・ K)
A: Given :- ∆G° = -237.2 kJ/mol Temperature (T) = 298 K R = 8.314 J/K. mol To calculate :- value of…
Q: If ΔG0 = −106 kJ/mol for this reaction at 298K, what is the Keq at this temperature? Recall: R =…
A: The relation between ∆G° and equilibrium constant,Keq,is given by the equation :…
Q: What is the free energy change for the following reaction at 25 oC if the entropy change at this…
A: The reaction given is 3 A (g) + B (s) ------> 2 C (g) + 3.40 kJ Since heat is written in product…
Q: Vhat is the standard free-energy change, AGº, for the following reaction at 44.5 °C? N2(g) + 3H2(g)…
A: For any reaction, the standard free energy change is given by ΔG0 = ΔH0 - TΔS0 And since the ΔH0…
Q: the K for a reaction is determined to 3.81 at 20.26 ºC, then what is ΔG for the reaction (in J/mol,…
A: Given-> K = 3.81 T = 20.26 °C = 20.26 + 273 = 293.26 K
Q: Note: if you have not been given enough information to decide a sign, select the "unknov reaction…
A:
Q: 19-20. For each reaction at STP, what is the sign of AS° in the blank. 19. 2H2 (g) + O2 (g) → H20…
A: Entropy is the degree of disorderness of a system.in gas the entropy is maximum as gaseous molecules…
Q: spontaneous? One with a +AH and a -AS One with a -AH and a J S
A: We will choose Correct option
Q: For which reaction in each set will AS° be more significant? For which reaction will AS be positive?…
A: Entropy is a measure of the freedom of motion in a system. Restricting the freedom of motion of a…
Q: Calculate AH for the reaction of sulfur dioxide with oxygen. 2SO2 (9) + 02 (g) → 2SO3 (g) (AHF° SO2…
A: Enthalpy is a parameter of energy used to measure the energy in thermodynamic reactions. In…
Q: II. GENERAL INSTRUCTIONS Read and analyze carefully each problem. Show your complete solution. 1.…
A:
Q: What is the free energy change for the reaction below, if the reaction quotient, Q, at 25.0oC is…
A: The relation between ∆G and ∆Go at absolute temperature T is given by: ∆G = ∆Go + RTlnQ, where, Q is…
Q: What is the sign of Δ S for the reaction below as written? CaCO3(s) ---------> CaO(s) +…
A: We are to discuss what is sign of ΔS for reaction CaCO3(s) ---------> CaO(s) + CO2(g)
Q: Calculate Δ So for the reaction below? 4 Fe(s) + 3 O2(g) ---------> 2 Fe2O3(s)
A: Known: SoFe(s)=27.3 J/K·molSoO2(g)=205.0 J/K·molSoFe2O3(s)=87.4 J/K·mol
Q: What is the free energy change for the reaction below, if the reaction quotient, Q, at 25.0oC is…
A: We will calculate free energy change.
Q: Under standard conditions, how much energy can be produced (i.e. what is ΔG'o ) when 1 mole of NADH…
A: Half cell reaction is given by, NaDH→NaD++2e-+H+ E0=0.320 V12O2+2e-+2H+→H2O E0=0.817 V…
Q: Explain how the value of ΔH for a reaction changes upon each operation.a. multiplying the reaction…
A: In a chemical reaction, delta H represents the sum of the heats of formation of the products minus…
Q: Calculate the value of ∆H for the following reaction given this additional information; Al (s) +…
A: The expression for change in enthalpy is shown below: ∆H=Σn∆Hfproducts -Σm∆Hfreactants where;…
Q: Which of the following statements determine a reaction to be spontaneous? O The ASpxn is positive. O…
A: The relation between change in entropy, enthalpy, and Gibbs free energy can be given as: ∆G=∆H-T∆S…
Q: What is K for a reaction if ∆G° =-216.0 kJ/mol at 25°C (or 298 K)? (R = 8.314 J/mol ・ K)
A:
Q: Calculate ∆S° values for the following reactions by using tabulated S° values from Appendix C. In…
A: Since you have posted multiple sub parts, the answer for first three subparts are given below.…
Q: Estimate ΔG° rxn for the following reaction at 449.0 K. CH 2O( g) + 2 H 2( g) → CH 4( g) + H 2O(…
A: Given :- CH 2O( g) + 2 H 2( g) → CH 4( g) + H 2O( g) ∆H° = -94.9 kJ ∆S°…
Step by step
Solved in 2 steps with 2 images

- At 381 K, the following reaction takes place: Hg2Cl4(g) + Al2Cl6(g) --> 2 (HgAlCl5)(g) ΔH = +10 kJ (a) Offer an explanation for the very small ΔH for this reaction for the known structures of the compounds. (img 1) (b) Explain why the small ΔH in this reaction is evidence against (img 2) as the structure of Hg2Cl4(g).Using S° values from Appendix C, calculate ∆S° values for the following reactions. In each case, account for the sign of ∆S°.(a) C2H41g2 + H21g2 ¡ C2H61g2(b) N2O41g2 ¡ 2 NO21g2(c) Be1OH221s2 ¡ BeO1s2 + H2O1g2(d) 2 CH3OH1g2 + 3 O21g2 ¡ 2 CO21g2 + 4 H2O1g2What is the free energy change for the reaction below, if the reaction quotient, Q, at 25.0oC is equal to 4.56e-6?2C2H2(g) + H2(g) ↔ C4H6(g) ΔGo = -216.790 kJ. ΔGo = -219.346 kJ ΔGo = 589.938 kJ ΔGo = -214.234 kJ ΔGo = -247.262 kJ
- Give good solution ASAP Thanks Which is more likely to be thermodynamically favored, the forward reaction or the reverse reaction? If C6H6(g) were consumed instead of C6H6(l), would you expect the magnitude of ΔHΔH to increase, decrease, or stay the same? The ____ reaction has a _______ ΔH value and, therefore, is more likely to be thermodynamically favored. If C6H6(g) were consumed instead of C6H6(l), magnitude of ΔH would ______, since in this case, the reactant is in the _______ enthalpy gas phase.Which of the following will produce a spontaneous reaction? (deltaG < 0) - deltaH < 0 deltaS < 0 low temp -deltaH < 0 deltaS > 0 all temp -delta H > 0 deltaS > 0 low temp -deltaH > 0 delta S <0 all tempCalculate ΔG°rxn, ΔH°rxn, and ΔS°rxn of the following reaction: 2O3(g) -> 3O2(g)
- Comment on whether ΔH and ΔS favor spontaneity of the following reactions. Explain your reasoning!a) Ba(OH)2.8H2O (s) + 2NH4Cl (s) BaCl2 (aq) + 2NH3 (g) + 10H2O (l) ΔH = + 80.3 kJΔS = + 428 J1. Predict the sign of ∆S for the following reaction. a. negativeb. positivec. no change 2. What is an intermediate? a.A local maximum on the energy diagram. b. A point on the reaction pathway that has a discrete minima. c.A point half-way between the starting materials and products. d. The highest energy compound on an energy diagram.Compare all four energy diagrams. Which one exhibits the largest Ea and which processes will have a value of Keq that is greater than 1?
- 40 Compute the standard free energy change for the following reaction: CaSO4(s) + 2HCl(g) → CaCl2(s) + H2SO4(l)What is ∆G° for the reaction CH₃OH(g) → CO(g) + 2 H₂(g) at 25°C?Predict the signs of rHo and rSo for the transformation: CH3OH(l) ->CH3OH(g) A.) rHo > 0 and rSo > 0 B) rHo < 0 and rSo > 0 C) rHo > 0 and rSo < 0 D) rHo < 0 and rSo < 0

