2. Roughly sketch the hydropathy plot for the given hypothetical membrane protein. Follow the color assignments of the helical domains and properly label your plot and axes (NB: Only a rough sketch is being asked. No need be exact with the hydropathy indices and residue numbers).
2. Roughly sketch the hydropathy plot for the given hypothetical membrane protein. Follow the color assignments of the helical domains and properly label your plot and axes (NB: Only a rough sketch is being asked. No need be exact with the hydropathy indices and residue numbers).
Anatomy & Physiology
1st Edition
ISBN:9781938168130
Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Chapter3: The Cellular Level Of Organization
Section: Chapter Questions
Problem 6RQ: Because they are embedded within the membrane, ion channels are examples of ________. receptor...
Related questions
Question
Answer the given question Clearly and Graph it neatly
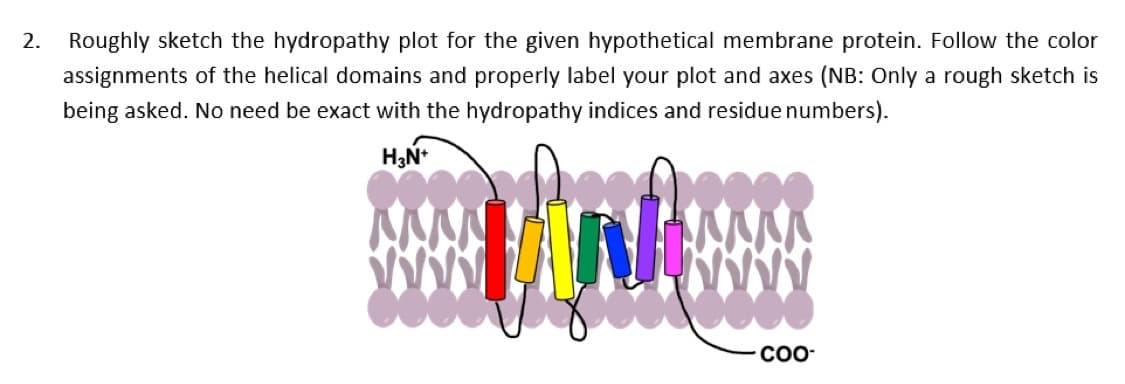
Transcribed Image Text:2. Roughly sketch the hydropathy plot for the given hypothetical membrane protein. Follow the color
assignments of the helical domains and properly label your plot and axes (NB: Only a rough sketch is
being asked. No need be exact with the hydropathy indices and residue numbers).
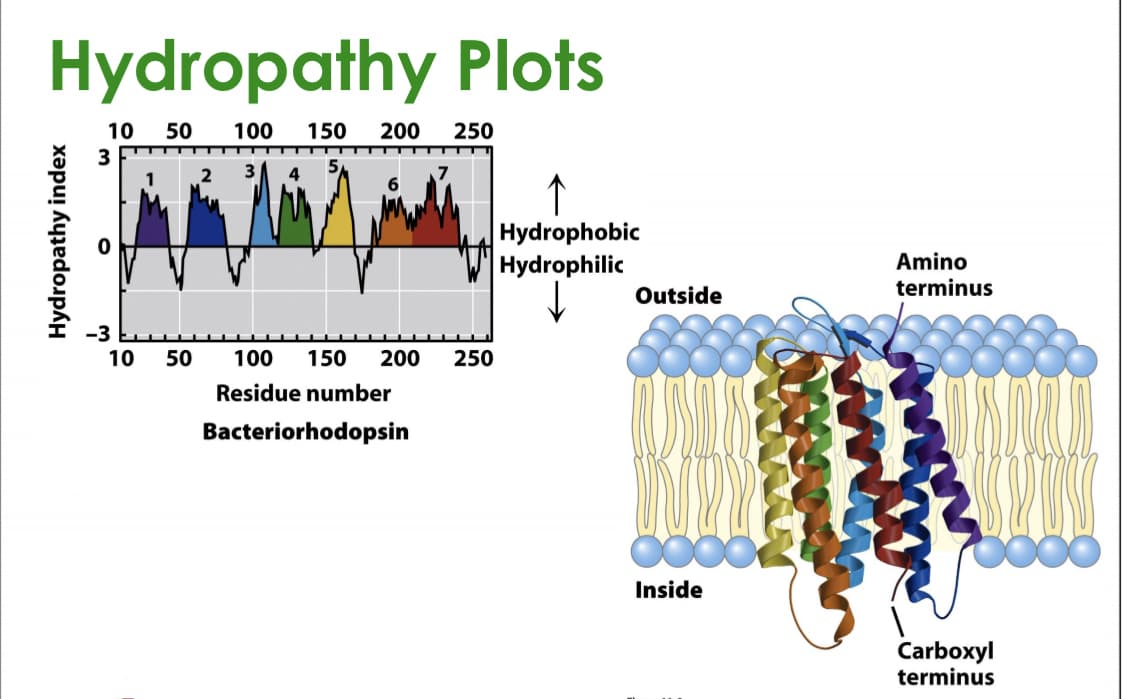
Transcribed Image Text:Hydropathy Plots
10
50
100
150
200
250
3
↑
Hydrophobic
Hydrophilic
Amino
terminus
Outside
-3
10
50
100
150
200
250
Residue number
Bacteriorhodopsin
Inside
Carboxyl
terminus
Hydropathy index
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax