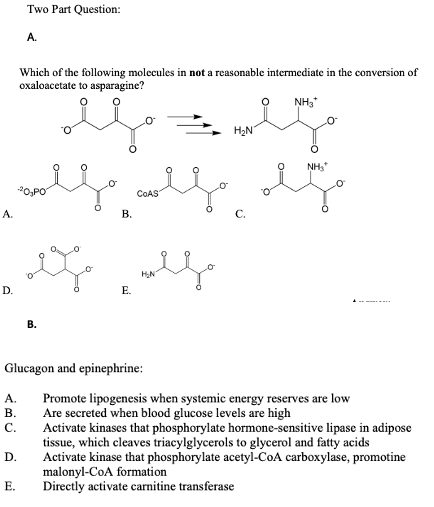
A. Which of the following molecules in not a reasonable intermediate in the conversion of oxaloacetate to asparagine? NH, H2N NH, O,PO COAS A. В. C. HN D. Е.
Q: Which factor is least likely to result in protein denaturation? disruption of weak interactions by b...
A: Protein denaturation is the process by which a protein loses its tertiary structure and therefore it...
Q: Draw and name three different functional groups. Draw the basic structure of an amino acid. Show how...
A:
Q: What is meant by the statement “The genetic code is universal”? What is the significance of this fin...
A: DNA is deoxyribonucleic acid that contains genetic information. Gene is a segment of DNA that can pr...
Q: The following results were obtained for an enzyme-catalysed reaction Substrate concentration (mmol l...
A: Enzymes are highly specialized proteins that have extraordinary catalytic power, greater than that o...
Q: What is it about the structure of cellulose and starch would cause these two sugars to produce lower...
A: Carbohydrates are classified as monosaccharides, oligosaccharides, and polysaccharides based on the ...
Q: hospital, his physician told him that his infarction is the result of a long history of smoking. Exp...
A: Cholesterol is an important steroid required for many steroid synthesis like testosterone. It is pre...
Q: A researcher in Biochemistry is isolating angiotensin-converting enzyme (ACE) from rabbit lung tissu...
A: Introduction: A buffer solution is an aqueous solution that contains a mixture of a weak acid and it...
Q: Amino acids are classified according to the R group linked at the carboxyl group. a. True b. False
A: Amino acids are the monomeric units of the proteins. Generally , 20 types of amino acids are found i...
Q: Provide differences between solution-phase peptide synthesis and solid phase synthesis in terms of: ...
A: Now a days small peptide is synthesized and tested its drug property to treat a complex disease or d...
Q: State Two points to support the statement below, using adequate and sufficient information to clearl...
A: Humans have 13 vitamins, which are categorized based on their solubility. Nine vitamins are water-so...
Q: What amino acids are present on the unknown sample based on the results below? Test/s Results Ninhyd...
A: There are several test, which are used to perform in order to identify the specific amino acid in a ...
Q: Given the chromatogram below in a normal phase, which samples would be the least polar? O Samples Ba...
A: In chromatography, for comparing relative polarity of the molecules, the retardation factor (Rf) ca...
Q: The protein portion of a conjugated enzyme is called a(n).
A: Enzymes are proteins that facilitate in the speeding up of our bodies' metabolism, or chemical react...
Q: What determines the fluidity of the biomembranes? Stiffness and rigidity of biomembranes depends on ...
A: Biological membranes are the chemical composition of phospholipid bilayer with peripheral and integr...
Q: In cation exchange chromatography, positively charged proteins exit the column first. In anion excha...
A: Chromatography is a technique in which components of a mixture are separated. It consist of a mobile...
Q: Which statement about membrane lipids is TRUE? Question 7 options: Glycerophospholipids are f...
A: Membrane lipids- Lipids that form the lipid-layer in the cell membrane
Q: Determine the catalytic power of jack bean urease in catalyzing the hydrolysis of urea given the fol...
A: ENZYMES : - Enzymes are defined as the organic molecules which helps in catalyzing the reaction....
Q: IX. Insulin, a hormone vital in blood sugar regulation and having a polypeptide chain with disulfide...
A: Insulin is a peptide hormone that downregulates blood glucose levels. Hormone insulin contains two p...
Q: -luginfile.php/7382296/mod_resource/content/0/Purine%20Practice%20Pathway.pdf 1 / 1 67% Purine Synth...
A: DNA and RNA are nucleic acids composed of nucleotides. Nucleotides are composed of a pentose sugar (...
Q: What is the best interpretation from the results?
A: 1. Negative ninhydrin test shows that the sample does not contain free amino acids. 2. Positive xant...
Q: What is the term used to refer to the functional, folded conformation of proteins? Natural O Native ...
A: Proteins are the made up of several units of Aminoacids joined by peptide bond. There are 4 levels o...
Q: Peptide Concentration (M) 2.5 x 104 5.0 x 104 10.0 x 104 15.0 x 104 Velocity (M/min) 2.2 x 106 5.8 x...
A: Km is the substrate concentration at half vmax.vmax is the maximum rate of reaction catalysed by Enz...
Q: Monosaccharide units are hydrophilic and generally soluble in water. What is meant by the statement?...
A: Monosaccharides are carbohydrates that cannot be broken down further by hydrolysis into simpler or s...
Q: An amino acid structure contains a -COOH and -NH2 group in the same molecule. The -COOH group donate...
A: The ability of the amino acid to exhibit both positive and negative charges causing the amino acid t...
Q: What chemical is used to break disulfide bonds, preliminary to detection of cysteine residues in a p...
A: Disulphide bond:- 2 cystine residues oxidises and form a covalent bond ( bridge ) between 2 sulphur...
Q: CH-OH НО OH What is the name of this structure? OH OH alpha-D-gulopryranose O beta-L-gulofuransone O...
A: Pyranose : Has 6 membered ring with 5 carbon atoms and 1 O atom Furanose : Has 5 membered ring with ...
Q: II. Differentiate the following in terms of proteins' structural features in living systems. 1. prim...
A: The structure of a protein are organized at different levels of primary, secondary, tertiary and qua...
Q: Modified TRUE or FALSE. Write the word TRUE if the statement is correct. If the statement is false, ...
A: Proteins are made up of amino acids, which are a type of molecule. The basic components of life are ...
Q: In Chapter 3, the Kd of calmodulin EF hands for binding Ca2+ is given as 10−6 M. Many proteins have ...
A: Calmodulin is a Ca2+ sensor with EF-hands as its high-affinity Ca2+ binding domains, making it a pro...
Q: The detection of single nucleotide polymorphisms is possible with: O A. Restriction Enzymes O B. All...
A: SNPs or single nucleotide polymorphisms are the most common type of genetic variation in humans. Th...
Q: A protein you want to isolate is often associated with the cell membrane. After homogenizing your sa...
A: Ammonium sulphate is often used to either solubilize or precipitate the protein depending on what co...
Q: MULTIPLE CHOICE 1. High density lipoproteins is an example of which kind of protein? A. Regulatory...
A: Proteins are composed of twenty standard amino acids attached together via peptide bonds. These twen...
Q: Prolamins are proteins soluble in water and dilute aqueous solutions
A: With respect to the solubility of a protein in water the globular proteins are more soluble than fib...
Q: 1. Based on your knowledge about gums and mucilages, how will you differentiate resins from them? (O...
A: Q. 1. Mucilages are generally normal products of metabolism (physiological products), formed within ...
Q: Which of the following is the most likely explanation for a situation where the ELECTRON TRANSPORT C...
A: The electron transport chain is made up of membrane-embedded proteins and organic compounds that are...
Q: Assigned Member of the Group of Alcohol Dehydrogenase and Lysozyme
A: Alcohol dehydrogenase is an enzyme that catalyses the conversion of ethanol to acetaldehyde. CH3-CH2...
Q: why chromotography isn't a good fit to detemine mitochondrial RNA polymerase
A: Mitochondrial RNA polymerase (mtRNAP) is vital for biogenesis of mitochondria as well as mitochondri...
Q: It is the main antioxidant used by the body to keep hemoglobin in its reduced form
A: The most common type of hemoglobin in adult humans is in the form of a tetramer that contains two no...
Q: A linear plasmid contains only two genes, which aretranscribed in opposite directions, each one from...
A: Plasmids are small, circular DNA molecules found in bacteria and other organisms. Plasmids are indep...
Q: Which of the following differentiates amylose with cellulose? The two polysaccharide have different ...
A: Amylose : It is a storage polysaccharide Cellulose : It is a structural polysaccharide
Q: Modified true or false. Write the correct answer if the underlined word is false.
A: Any organic molecule that consists of a basic amino group, an acidic carboxyl group, and a distinct ...
Q: Assume that a certain cell, the ratio products/reactants or Keq 560.6 (Keq dimensionless) reaction G...
A: A reversible reaction is one that can go both forward and backward. When the rate of the forward rea...
Q: Differentiate fertilized and unfertilized egg of Ascaris. Differentiate male and female worm of Ancy...
A: Introduction: Ascaris lumbricoides is a roundworm of man and it is the largest intestinal nematode p...
Q: At high salt concentration, proteins are less soluble. Changing the solubility of a protein based on...
A: Salting out procedure can be used to precipitate large biomolecules, such as proteins or DNA. The sa...
Q: Amylopectin and cellulose are being compared. Which of the following choices explains the similarity...
A: Carbohydrates are composed of carbon, oxygen, and hydrogen which are connected by the glycos...
Q: Experimental results describing a protein's amino acid composition are useful for estimating the mol...
A: Proteins are biological macromolecules composed of amino acids linked together by peptide bonds. The...
Q: Given a tripeptide Cys-His-Lys, Cys: Pk1 = 1.71; Pk2 = 10.78; PkR = 8.33 His: Pk1 = 1.82...
A: Hi. Thank you for the question, As per the honor code, we are allowed to answer three sub-parts at ...
Q: Why is the peptide formation always presented/read from N-terminal to C-terminal?
A: Introduction: Amino acids are joined together by a condensation reaction between carboxylic and ami...
Q: An attempt was made to inhibit Enzyme X by adding compounds M and L. The results are summarized in t...
A: Inhibitors are substances which decrease the enzyme activity by inhibiting the enzyme. Inhibitors ar...
Q: Which is responsible for the stabilization of parallel polypeptide chains in a beta-pleated sheet co...
A: Secondary structure refers to the local folded structures that occur inside a polypeptide as a resul...
Step by step
Solved in 8 steps

- The drug troglitazone was used to treat diabetes but was withdrawn from the market when patientswho took the drug suffered from severe side effects. The data below show the activity of an enzymein the steroid biosynthetic pathway in the presence and absence of 10 µM troglitazone.[S] (µM) v0 (pmol min-1)Create a plot of 1/v0 vs. 1/[S]. Calculate KM and vmax in presence and absence of inhibitor. Whattype of inhibitor is troglitazone? Clearly support your answer.Lithium ion, which is used to treat bipolar disorder, interferes with the phosphoinositide signaling pathway by inhibiting enzymes such as inositol monophosphatase and inositol polyphosphate 1-phosphatase. Predict the eff ect of Li+ on the supply of cellular inositol, a precursor of phosphatidylinositol and PIP2.With the aid of diagrams describe the signalling pathway involving inositol 1,4,5 trisphosphate from ligand binding to intracellular effects (65%). In your answer explain how it is synthesised from the membrane phospholipid and, excluding receptor agonists, provide two examples of drugs that modulate activity in the pathway (35%).
- One such laboratory study investigated the binding of a hormone to three different receptor proteins in the cell membrane. The data collected are shown in the table below 1) Provide a brief explanation as to why ligand binding toproteins must be a reversible process. 2) Calculate the dissociation constant (Kd) for the hormone binding to each of the three proteins.260. A 42-year-old woman is found to have type 2 diabetes mellitus. The physician recommends treatment with exenatide, a glucagon-like peptide-1 (GLP-1) receptor agonist. Which of the following is most likely to occur as a result of the effect of GLP-1 on glucose homeostasis in the patient?a.Glucagon secretion in response to fastingB. Glucagon secretion in response to a mealC. Insulin secretion in response to fastingd. Insulin secretion in response to a mealDetermine the effect (either no effect, increase, or decrease) of the following: a. What is the effect of high cholesterol in the LDL receptor synthesis? Explain. b. What is the effect of intaking statin on the concentration of squalene? Explain.
- The metabolic precursor for the production of most eicosanoids isOne such laboratory study investigated the binding of a hormone to three different receptor proteins in the cell membrane. The data collected are shown in the table below: 1) Provide a brief explanation as to why ligand binding to proteins must be a reversible process. 2) Calculate the dissociation constant (Kd) for the hormone binding to each of the three proteins. 3) Which of the proteins demonstrate the tightest/strongest binding of the hormone? Which demonstrates the least tight binding of the hormone? What is the basis for your answer?Explain in detail the funcations and signalling and activation of ephrin receptor EPHB2. Give step by step explanation
- BIOC 384 G Protein-Coupled Receptor (GPCR) Signaling Q.7.3: Glucagon and epinephrine both signal stress, which would be low blood glucose levels or acute danger, respectively. Describe the upstream signaling pathways that would be activated in liver cells if you were hungry from a long hike in Sabino Canyon and came across the biggest rattlesnake you had ever seen right in front of you on the trail.The epinephrine-mediated “amplificationcascade” of Figure 24.14 has six steps, all of which are catalyticwith one exception. This cascade leads to the activation of glycogenphosphorylase. This enzyme acts in turn on glycogen to yieldglucose-1-phosphate (G-1-P).(a) Which step is not catalytic?(b) If each catalytic step had a turnover (molecules of substrateacted on per molecule of enzyme) of 10, how many moleculesof G-1-P would result from one molecule of epinephrine?(c) What is the biochemical advantage of such a cascade?(d) How is the amplification cascade of this questionreversed?Provide a diagram of the EPH RECEPTOR B2 (EPHB2) structure. Give annotations of binding sites and enzyme active sites.

