Q: One step in the synthesis of the antihistamine fexofenadine (Section 23.5) involves acid-catalyzed…
A: Given Structure: Reaction of A in presence of acid-calatysed hydration of H+ Following products are…
Q: 'N: `CH3 H3C
A: We have to carry out the given transformation.
Q: 11. Identify the reagent necessary to accomplish the following transformations:
A: Here, the first and second step involves the stepping up of carbon. This can be done by treating…
Q: 1a). What two alcohols could give the following Allende upon treatment with concentrated sulfuric…
A: Alcohols on reaction with sulphuric acid undergo dehydration to form alkenes.
Q: Show how you would use the Williamson ether synthesis to prepare the following ethers. You may use…
A: We are authorized to answer three subparts at a time, since you have not mentioned which part you…
Q: 3. Draw the product of the reaction of propanal with each of the following reagents: a. Lithium…
A: Lithium aluminium hydride is a strong reducing agent.It reduces aldehyde into alcohol.
Q: AOC XT H3O+ H H OH OH Epoxides are cleaved by acid as are other ethers, but milder conditions are…
A:
Q: One step in the synthesis of the antihistamine fexofenadine involves acid-catalyzed hydration of the…
A: Acid catalyzed hydration of alkynes- Acid catalyzed hydration of alkynes produce ketone.…
Q: 4. (A) Give the reagent that would be required to carry out the following transformations. Br
A: (I)AcetyleneNaNH2 (II)H2/Pd (III)OsO4/NaHSO3(I) (II) (III)AcetyleneNaNH2H2/PdOsO4/NaHSO3…
Q: 2.11. What is the best description of reactions to carry out that will arrive at the following…
A: Synthesis of aldehyde using the right steps.
Q: How can you prepare the following compounds with benzene as one of the starting materials?
A: a) The given compound is Diphenyl. Chlorination of benzene in the presence of catalyst anhydrous…
Q: Draw the structure/s of the product/s of the reaction of the following reagents with toluene (2…
A: Friedals craft alkylation: these are the reactions in which alkyl group is added to benzene and its…
Q: Below are two potential methods for preparing the same ether, but only one of them is successful.…
A: 1- In First reaction ( which is extremely left side ) is successful approach means given ether in…
Q: 7. Compound X is an alcohol. Upon oxidation by acidify potassium permanganate, a ketone, Y is…
A: Since, you have asked multiple question, we will solve the first question for you. If you want any…
Q: A chiral ether of molecular formula C5H100 reacts with hot HI to give a product of molecular formula…
A: Molecular formula of reactant= C5H10O Molecular formula of product= C5H10I2
Q: 3. For each of the following, state whether it would undergo nitration faster, slower or at the same…
A:
Q: 7. Propose efficient synthesis for the following reaction. an
A:
Q: What alkyl halide reacts with lithium divinylcuprate 3(CH2“CH)2CuLi4 for the synthesis of each of…
A: The molecular formula of lithium divinylcuprate = C4H6CuLi (a) When bromocyclohexane is treated with…
Q: Suggest a synthesis of the following compound by Claisen or Dieckmann condensations. Show all…
A: It is Claisen Condensation reaction. It is a base catalysed reaction. Reactant is ester.
Q: [4] What is the major product obtained from reaction of acetone with cyclopentane-1,2 diol under…
A:
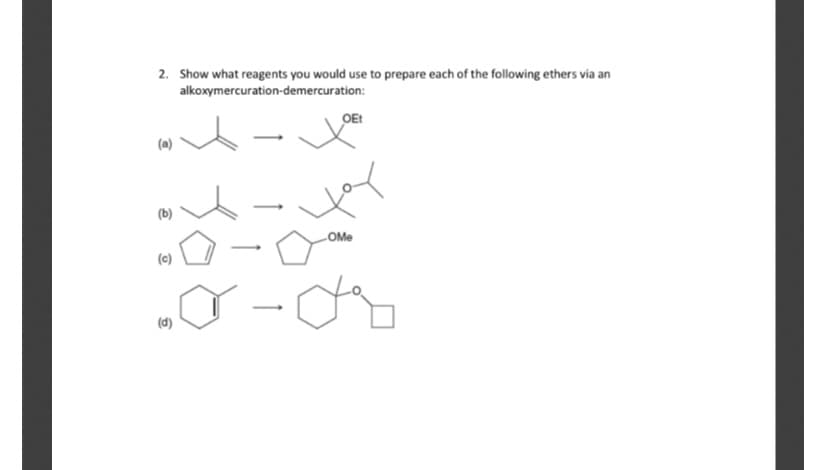
Q: Show how the following ethers might be synthesized using (1) alkoxymercuration–demercuration and (2)…
A: Given molecule: (a) 2-methoxybutane
Q: 3. What is of the following compounds will produce a mixture of constitutional isomers when treated…
A: Unsymmetrically disubstituted alkene/alkyne upon reaction with HBr give a mixture of constitutional…
Q: 36. The ring-opening of cyclic ethers happens through cleavage by suitable nucleophiles. Which among…
A: The bond angle of the tetrahedral carbon atom (sp3 hybridized carbon atom) is 109o28'. In the…
Q: c) d)
A:
Q: Identify the reagents necessary to achieve each of the following transformations: он он (a) (b) (c)
A:
Q: 3. (a) Strategies the synthesis of the given transformations to ensure no side-product (provide the…
A:
Q: Q12. (LkrAmostby)benzene 1 ydergoes an elimination following an E1 mechanism. Fill in the following…
A:
Q: b) What starting materials are needed to prepare the compound below by intramolecular Mic reaction?…
A: The answer is given as follows
Q: Explain briefly by illustrations the chemistry behind each answer 1. Preamble :A reaction flask…
A: Given: 2-bromopentane in ethanolic solution of sodium ethoxide forms 1-pentene and 2-pentene.
Q: CI I II IV
A: The correct answer is given below
Q: 2. For the following structure: (1S, 2R, 4R)-2-chloro-1-isopropyl-4-methylcyclohexane c. Draw the…
A:
Q: 8. Provide the reagents necessary to prepare the following compound using a Williamson ether…
A: phenyl propylether is synthesised by the reaction of phenol and propyl iodide in the strong alkaline…
Q: how how you would synthesize the following compounds, starting with benzene or toluene and any…
A: Given preparation,
Q: Suggest a synthesis of the following compound by Claisen or Dieckmann condensations. Show all…
A: Dieckmann condensation is the intramolecular (within the molecule) condensation of diester compound…
Q: Fill in reagents A, C, E and compounds B, D in the following transformation. A В C D E CI.
A:
Q: b) Provide the synthons and synthetic equivalents for the following compounds. i) Synthons:…
A:
Q: 7. The addition of organometallic compounds to carbonyls is one of the reactions of great interest…
A: Interpretation: To complete the synthesis route and perform reaction mechanism of formation of B.…
Q: 9. (a) Provide the reagent necessary to carry out the following chemical transformations. (i)
A: Here we have predict the suitable reagents required for the following given conversion .
Q: Explain the reactivity and orientation effects observed in each heterocycle. a. Pyridine is less…
A: Aromatic compounds like benzene show some specific chemical reactions like electrophilic…
Q: Using any alkene and any reagents necessary, how would you prepare the following compound?
A: Coversion of an alkene to the given alcohol is as follows.
Q: Propose an efficient and elegant synthesis of the following molecule starting with benzene. You may…
A:
Q: 3. Propose a structure for an alkyl halide that generates only (Z)-3-methyl-2-phenylpent-2-ene upon…
A: The alkyl halides can be converted to an alkene with the help of a base. This reaction is called…
Q: 3. (a) Strategies the synthesis of the given transformations to ensure no side-product (provide the…
A:
Q: Rank the compounds in each group in order of increasing reactivity towards nucleophilic addition a)…
A: An electrophile is a carbon species that might be neutral or positively charged. Similarly, a…
Q: Identify the reagents "a" in the following scheme: OH CH3 CH3 CH3 b. NaOCH3 with acid @ low…
A: In the elimination reaction, the formation of alkene takes place. there will elimination of a proton…
Q: The following compound is undergo ether cleavage with strong acid. OCH3 2 HBr N + P i) Draw the…
A: Given : We have to make products for the following reaction.
Q: How you would prepare the following compound from toluene and any other reagents? ČH3
A:
Q: Give the relative rates of reaction of propyl acetate with the four reagents below to give a…
A: Reactivity depends on the polarity of bonds. The higher the polarity, the more will be the…
Q: 4. Propose an efficient synthesis for each of the following transformations. (d) (e)
A: Interpretation - To propose an efficient synthesis root for each of the following given…
Can you help me with this problem
When alkene reacts with mercuric acetate in a THF solvent, Mercurinium ion is formed. Which can be attacked by nucleophile where the possible carboncation will have higher stability. In next step the acetoxymercury group can we reduced by alkaline sodium borohydride.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- 16. When cis-1,2-cyclopentanediol reacts with acetone in dry HCl, compound M is formed. What do you expect the hydrolysis of compound M to be when subjected (i) to base; and, (ii) acid A. resistant to base; resistant to acid B. resistant to base; readily hydrolyzed by acid C. readily hydrolyzed by base; resistant to acid D. readily hydrolyzed by base; readily hydorlyzed by acidIdentify the reagents you would use to achieve of the following transformation. Identify these reactions as SN1 or Sn2. (a) (b) (c) (d)16. When cis-1,2-cyclopentanediol reacts with acetone in dry HCl, compound M is formed. What do you expect the hydrolysis of compound M to be when subjected (i) to base; and, (ii) acid a. resistant to base; resistant to acid b. resistant to base; readily hydrolyzed by acid c. readily hydrolyzed by base; resistant to acid d. readily hydrolyzed by base; readily hydorlyzed by acid 17. Following (16), would you expect that a similar compound be formed if the reactant was trans-1,2-cyclopentanediol? Why is this so? a. Yes; hydroxyl groups are on opposite side b. No; hydroxyl groups are on opposite site c. Yes; hydroxyl groups are on the same side d. No; hydroxyl groups are on the same side
- 1. What effect would you expect on the rate of reaction of ethanol with 2-iodo-2-methyl-butane if the concentration of the halide is tripled? 2. Explain the difference between addition, elimination and substitutionreactions. 3. Optically active 2-butanol slowly racemizes on standing in dilute sulfuricacid. Propose a mechanism to account for this observation.v) What products would you expect if 2-butyne were treated with ozone in the presence ofacetic acidvi) What products would you expect if 1-butyne were treated with ozone in the presence ofacetic acidvii) What products would you expect if 2-butyne were treated with KMnO4 in the presenceof KOH in water?viii) What products would you expect if 1-butyne were treated with KMnO4 in the presence of KOH in water1.Describe the ozonolysis of alkenes 2.one mole of a hydrocarbon(A) reacts with one mole of beomine giving a dibromo compound C5H10Br2.Substance A on treatment with cold dilute kMnO4 solution forms a compound C5C12O2(C5H12O2) on ozonolysis A,gives equimolar quantities of propanone and ethanol.Deduce the structure of substance A.
- Explain briefly by illustrations the chemistry behind each answer 1. Preamble :A reaction flask contains a 2-bromopentane in an ethanolic solution of sodium ethoxide at room temperature and results in the formation of two olefinic products(1-pentene and 2-pentene) I) What reaction pathway is followed by the reaction above?A. E2 dehydrohalogenation B. E1 dehydrohalogenationC. SN1 dehydrohalogenationD. SN2 dehydrohalogenationE. A mixture of E1 and E2 pathways II) What is responsible for the formation of different products (major and minor).A. The different activated complex involved in the mechanism.B.Bimolecular Nucleophilic substitution reaction C.Bimolecular Elimination reaction D.The presence of sodium ethoxideE.The hybridisation nature of the secondary carbocation III) All the following is true about the reaction in question 1 except?A. The reaction follows Zaistev’s ruleB. Sodium ethoxide is the nucleophile in this reactionC. The more highly substituted alkene is the most stable…7.56 Below are two potential methods for preparing the same ether, but only one of them is successful. Identify the successful approach and explain your choice.Provide a stepwise synthesis for benzyl alcohol to sodium benzoate with reagents KMnO4 and NaOH (including arrow-pushing mechanisms) Please show all the arrow push with the given reagents above.
- ) Draw all the Kekulé structures of anthracene and phenanthrene.(b) Propose mechanisms for the two additions shown.(c) In Chapter 8, most of the additions of bromine to double bonds gave entirely antistereochemistry. Explain why the addition to phenanthrene gives a mixture of synand anti stereochemistry.(d) When the product from (c) is heated, HBr is evolved and 9-bromophenanthrene results.Propose a mechanism for this dehydrohalogenationBelow are two potential methods for preparing the same ether (Option A and B), but only one of them is actually successful. Identify the successful approach (A or B)Show how you would synthesize the following phenols, using benzene or toluene as yourstarting material and explain why mixtures of products would be obtained in some cases.a. p-nitrophenolb. 2,4,6-tribromophenolc. m-cresol



