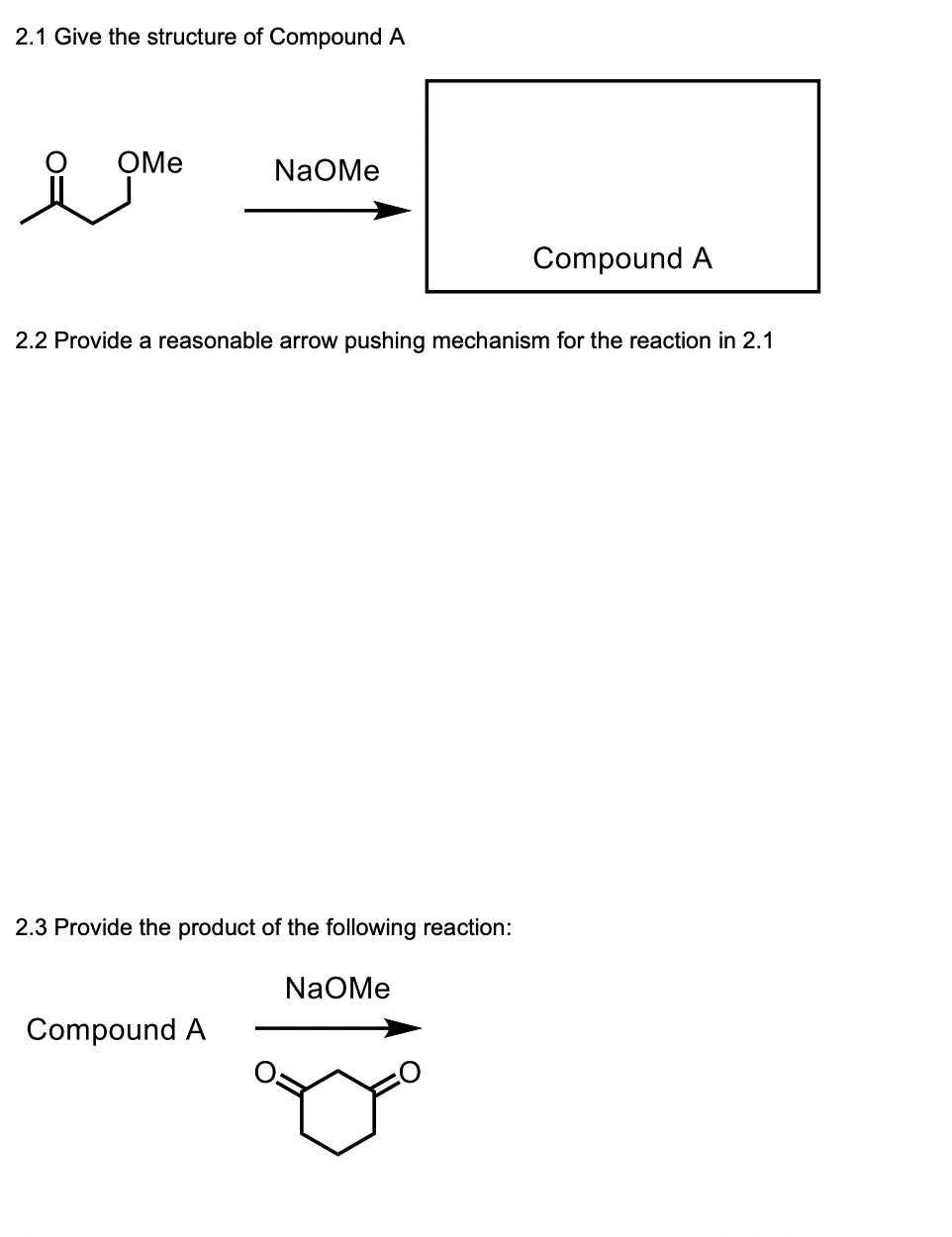
2.1 Give the structure of Compound A OMe NaOMe 2.2 Provide a reasonable arrow pushing mechanism for the reaction in 2.1 2.3 Provide the product of the following reaction: Compound A Compound A NaOMe
Q: Coal gasification produces an easily-transportable, clean-burning combination of carbon monoxide and…
A:
Q: What does Kp mean? Kp = gorganic layer gwater layer all choices are correct it is the equilibrium…
A: The objective of the question is to understand the meaning and interpretation of Kp in the context…
Q: [Review Topics] [References] Draw the structure(s) of the major organic product(s) of the following…
A: We have given the organic reaction and we have to find out the major product of the reaction.
Q: Some measurements of the initial rate of a certain reaction are given in the table below. [N₂] [H₂]…
A: rate=k[N2]2[H2]2k=1.9 M−3⋅s−1Explanation:
Q: 60. Write balanced equations for the complete combustion of each of the following substances. a.…
A: Combustion reactions involve the burning of a substance in the presence of oxygen thus hydrocarbon…
Q: 1) You are asked to prepare a buffer for a protein assay. This protein works best at a slightly…
A: A buffer is a solution which resist any change in pH on adding a small amount of acid or base.it is…
Q: Give me detailed Solution with explanation needed with structure..don't give Handwritten answer
A: The objective of this question is to determine the absolute configuration (R or S) of a chiral…
Q: Assign the absolute configuration of the chiral center as R or S. но- A. R BS OH C. R and S are both…
A: Given that, the structure of the molecule is:
Q: Provide the major product(s) for the reactions below
A: Given are organic reactions. The given reactions are oxidation reactions.Chiral compounds are…
Q: Complete the following table to demonstrate your understanding of the structures, IUPAC names, and…
A: The aldehyde (RCHO) gets oxidized to carboxylic acid (RCOOH) on reaction with oxidizing agents like…
Q: Do not give handwriting solution. Draw the curved arrow mechanism depicting the acid - catalyzed…
A: Product of following reaction can be made by applying appropriate reaction mechanism.
Q: Consider the equilibrium system described by the chemical reaction below. Calculate the value of Qc…
A: Given ->Weight of C = 11.1 gmWeight of H2O = 2.45 gmWeight of CO = 15.3 gmWeight of H2 = 7.55…
Q: EtO HO H* concentrated KMnO4 reflux OEt OH NaOCH3 OH 11 CrO3 H* 6 4 8 3 HCI, H₂0 1. NaH 2. HCI, H₂O…
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: -NH₂ 2 -NH₂
A: Pyridine-2-amine( structure 1) and pyridine-3-amine( structure 2) are both aromatic amines, which…
Q: A very simple IR spectrum of a drug does not contain bands above 3000 not any significant bands in…
A: Infrared spectra (IR) is used to determine the functional group present in the given compound.Each…
Q: 3. The results of an EAS iodination on indole are shown below. Note that this does not require an…
A: When all the properties of a molecule cannot be explained by a single structure, we draw more than…
Q: Which radioisotope produces 209 Bi via beta decay? 83
A: Since,In beta decay process loss of electron takes place. Due to this decay, number of proton…
Q: 2) A) What mass of air is required to burn 1.0 kg of ethylene to completion. (stoichiometric) B)…
A: Combustion reaction is the burning of any substance in the presence of oxygen to produce energy in…
Q: What is the relationship between the following molecules (enantiomers, diastereomers, meso, or…
A: The following rules are used to assign the R/S configurations to the chiral centers.Identify chiral…
Q: Consider a gas obeying van der waals equation. Let one mole of the gas expands reversibly from…
A: The Van der Waals equation of state is an improvement over the ideal gas law to account for the…
Q: Which one(s) is/are aromatic? 1 1, 2 and 3 only 1 only 4 1 and 3 ого 2 3 4
A: Here, four different organic molecule is given and we have to identify which among them are…
Q: Name the major organic compound formed by the following reaction: OCH 3 OCH 3 acid (hydrolysis)
A: A ketal is a functional group in organic chemistry that consists of a carbon atom bonded to two -OR…
Q: MDMA (or "Ecstasy") is a widely used illegal stimulant. In the following molecule, highlight each…
A: A trigonal planar geometry is a shape in which the central atom occupies the center of the triangle…
Q: Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and…
A: Given question is related to writing the product formed by using a curved arrow to represent the…
Q: Determine the major thermodynamic and kinetic products in the following reaction. Br₂
A: For a particular reaction, if there are possibilities to form more than one products, then the…
Q: H2SO3 + LiOH = ? Could you please show in detail the molecular, ionic, net ionic
A: It is a Double Displacement reaction where one mole of aqueous Sulfurous Acid [H>2SO3] and two…
Q: My lab partner writes the following calculation in our lab notebook: ((43.3 degrees C) (1.8 degrees…
A: Answer:Temperature is a physical quantity that can be represented in different units. are two…
Q: Write the balanced dissociation equation for soild lead (II) sulfate in aqueous solution. If it does…
A: we have to write the balanced dissociation equation for solid lead(II) sulphate in aqueous solution.
Q: What is a major neutral organic product for the following reaction? O Y 11... || 1. Br₂, H₂O 2. NaH…
A: Bromonydrin formation is addition reaction of Br and OH across the alkene and the product is called…
Q: Draw acid-base equilibrium for sulfamethoxazole (pka 5.8), and suldapyridine (pka 8.4).
A: The objective is to draw the acid-base equilibrium of the given compounds, Acids are compounds that…
Q: Please don't provide handwriting solution
A: Answer:Atomic size is inversely proportional to the effective nuclear charge on valence electron and…
Q: Name these molecules
A: The given molecule is represented by the formula O=O. This is a simple representation of the…
Q: Consider 2.00 mol of an ideal gas with a constant-volume molar specific heat of 26.0 J/(mol K), an…
A: The objective of this question is to calculate the change in entropy (ΔS) of an ideal gas during a…
Q: Draw the Newman projections for one staggered and one eclipsed conformation of the molecule below,…
A: The dot represents the front carbon and the circle represents the back carbon.
Q: Which nuclide results from the neutron capture of 183Os? 184Os, 182Os, or 184Re
A: Given is a neutron capture reaction of .To find the nuclide resulting from this nuclear reaction.…
Q: What has the greatest relative binding energy? 1H, 59Co, or 238U
A: Relative binding energy or binding energy per nucleon for an element can be calculated using the…
Q: НО, Me N OH NH2 НО, Me `N ОН ОН
A: To propose a synthetic route for the given target molecule from the given starting material
Q: At equilibrium, the concentrations in this system were found to be [N₂] = [0₂] = 0.100 M and…
A: The equation given is,N2(g) + O2(g) 2NO(g)Equilibrium concentration of N2 in this system ,[N2] =…
Q: Assign R/S configuration for carbons 2, 3, and 4 in the following structure: H H₂ H-3 I 10=0 I OH…
A: To determine the R/S configuration of the given Fischer projection.
Q: Most stable nuclide? 40Ca, 4K, or 41Ca
A: The objective of the question is to identify the most stable nuclide.
Q: 1) Drow the H-NMR of molecule A and B in a 2:3 ratio label Peaks and integration дох B Yox
A:
Q: plain how Le Chatelier's principle is used to determine the direction a system at uilibrium will…
A: Le Chatelier's principle
Q: Determine the change in kinetic chain length if the concentration of monomer decreases by 6.5 and…
A: A polymer is formed by a chain process.The concentration of monomer decreases by 6.5The…
Q: Here is a graph of the molarity of glucose (C6H₁206) in a reaction vessel during a certain chemical…
A: Given data:The graph of concentration of vs. time.Instantaneous Rate change at specific point is…
Q: REACTION II: Br ... KOCH3 CH₂OH
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: What is the product, A, that would be obtained from the following reaction sequence? NINH, H₂O+ A.…
A:
Q: Provide the structure of the major organic product of the reactions belo b. CI 1.P(OET)3 2. BuLi 3.…
A: Wittig reaction is formation of the alkene when aldehyde or ketones are treated with phosphorus…
Q: What is the relationship between the following two compounds? O constitutional isomers O identical…
A: Isomers are the compounds having same molecular formula but different chemical formula.…
Q: How many signals would you expect in the ¹3C NMR spectrum of the following compound? юн
A: 13C-NMR spectroscopy is mainly used for the identification of carbon skeleton of an organic…
Q: Draw a structural formula for the substitution product of the reaction shown below. + Na SCH3 CH3CN…
A: SN2 reaction is the bimolecular nucleophilic substitution reaction in which the rate of the reaction…
Trending now
This is a popular solution!
Step by step
Solved in 1 steps with 1 images

- Compound K, L and M are three isomers with the molecular formula C5H10O.Compound K cannot be oxidized, while compound M and L can be oxidized.Oxidation of compound L with hot acidified potassium permanganate,KMnO4 solution yields 2-methylbutanoic acid. When treated with Iodoformreagent, yellow precipitation only occurs in compound K. Fehling test onlyyielded positive results for compound L and negative for compound M andK. Compound M is then reacted with hydrogen chloride, HCl produceschlorocyclopentane (i) Draw the structural formula of compounds K, L and M.Compound K, L and M are three isomers with the molecular formula C5H10O.Compound K cannot be oxidized, while compound M and L can be oxidized.Oxidation of compound L with hot acidified potassium permanganate,KMnO4 solution yields 2-methylbutanoic acid. When treated with Iodoformreagent, yellow precipitation only occurs in compound K. Fehling test onlyyielded positive results for compound L and negative for compound M andK. Compound M is then reacted with hydrogen chloride, HCl produceschlorocyclopentane. (ii) Name the type of chemical reaction of compound M when reacted withhydrogen chloride, HCl .(−)-Hyoscyamine, an optically active drug used to treat gastrointestinal disorders, is isolated from Atropa belladonna, the deadly nightshade plant, by a basic aqueous extraction procedure. If too much base is used during isolation, optically inactive material is isolated. (a) Explain this result by drawing a stepwise mechanism. (b) Explain why littorine, an isomerisolated from the tailower plant in Australia, can be obtained optically pure regardless of the amount of base used during isolation.
- Provide a stepwise synthesis for benzyl chloride to benzoic acid. (including arrow-pushing mechanisms) Please show all the arrow pushCompare the chemical structure of compound A with that of compound B. Answer it in a detailed way. Thank You.(a) Illustrate the following name reactions by giving example :(i) Cannizzaro’s reaction(ii) Clemmensen reduction(b) An organic compound A contains 69.77% carbon, 11.63% hydrogen and rest oxygen. The molecular mass of the compound is 86. It does not reduce Tollen’s reagent but forms an addition compound with sodium hydrogen sulphite and gives positive iodoform test. On vigorous oxidation it gives ethanoic and propanoic acids. Derive the possible structure of compound A.
- Give the stepwise arrow-pushing mechanism for the following reaction to produce the product with stereochemical outcome.Provide a stepwise synthesis for benzyl chloride to benzoic acid. (including arrow-pushing mechanisms)When the nitrogen-containing aromatic heterocyclic compounds 1 and 2 are treated with HCl, only 1 forms the hydrochloride salt, whereas compound 2 is unreactive. Provide an explanation for this observed reactivity.
- Propose a curved arrow mechanism to account for the formation of compound C.Suggest curved arrow mechanism for 1 from 2 and identify compound ACompound X was soluble in water and ether, and its aqueous solution turned litmus blue. It reacted with sodium to give a gas. The compound reacted with benzenesulfonyl chloride and base to give an insoluble product, which was unchanged with acidification. It reacted with nitrous acid to give a yellow solid. Compound A could be

