Q: From the given pool of choices, give the most possible identity of A and B. A > B AICI3 AICI3
A: We have given a organic reaction. We have to find possible identity of A and B The given reaction…
Q: Phs° 12 Br R 'Buo Os04 then H30° 2 1 NH3/H2O MCPBA
A:
Q: phHệ CH 3 سار اي منهم مفقل الے۔ ph left (H3 ciz right CH₂ OM OH ؟ ko
A: The acidic strength depends upon the stability of conjugate base.Stable the conjugate base more will…
Q: A. Characteristics: Describe the physical properties of the following carboxylic acids. Molecular…
A: The boiling point of a substance is a specific temperature at which it starts boiling. Every…
Q: For the the molecule represented below: A Но. C OH ОН В Rank the labeled acidic protons (H atoms)…
A: The proton which can easily be removed is most acidic and vive versa.
Q: Which of the following is the most basic molecule? 0. FC O. O -OH fz6 OH OH
A: Acidity and basicity are opposite to each other. As the acidity of a compound increases its basicity…
Q: A) Base 1 is EtONa and base 2 is KOC(CH3)3 B) Both bases are KOC(CH3)3 C) Both bases are EtONa D)…
A: Kinetically controlled product: Product which form according to rate of reaction and depends on…
Q: What is the conjugate base to H2PO−3H2PO3− ? a. HPO2−3HPO32− b. HPO−3HPO3− c. PO3−3PO33− d.…
A:
Q: :0 H,SO4 HO, + CH3OH + H30* Ph Ph OCH3 H,+,H +:,H :0-H Ph OCH3 OH Ph ÓCH3 ОН Ph OCH3 Ph HO. а. b. С.…
A:
Q: Box 1 and 2. box 1 answer choices: [conj. acid]>[conj. base],[conj. acid]=[conj. base], or [conj.…
A: Buffer is the solution which resist a change in pH when an acid or a base is added into it.
Q: но. HO. Br
A: Acid are those chemical species when dissolved in aqueous solution loses its proton and form…
Q: Which are diastereomers?
A: diastereomers given in step 2
Q: 2. Citric acid has the molecular formula H3CH507, which implies that citric acid has how many acidic…
A:
Q: Acid Names IUPAC HCN(aq) Classical Acid Names IUPAC HCIO2(aq) Classical Acid Name IUPAC CH3COOH(aq)…
A: IUPAC name is the systematic name given to a chemical compound International Union of Pure and…
Q: Acids give pink color with Methyl Orange. Select one: True False
A: We know that Methyl orange is a pH indicator frequently used in titration because of its clear and…
Q: 1. The correct number of a acidic protons in the following compounds: (a) (b) (c) (d) (e) (f) а. (a)…
A: The answer of the above question is given in step 2
Q: 5. Calculate A H for H2(g) + Cl, (g) → 2HCI (g) based on the reactions: AH = -176 kJ AH = -99.22 kJ…
A: Given, a) NH3 (g) + HCl (g) → NH4Cl (s) ∆H = -176 kJ b) N2 (g) + 3H2 (g) → 2NH3 (g) ∆H = -99.22 kJ…
Q: 2. Below is adenine, one of the four nucleobases which make up the alphabet for DNA and RNA. Is the…
A: Base : according to Lewis substance having the ability to give up a pair of electrons are called…
Q: the pic (yellow circle l). What is the above O means? Is it 5OH^- or 5 O^- H?
A: Oxidation means loss of electrons and reduction means gain of electrons.
Q: Which group's results are incorrect and what mistake did they most likely make? O a) Group B is…
A:
Q: What is the [H3O +] of a pH 8.39 (watch your sig figs)?
A: pH of a solution is defined as negative log is concentration of H+ ion. pH=-log[H3O+]
Q: 2-37 Rank the following species in order of increasing acidity. Explain your reasons for ordering…
A: According to the Arrhenius concept the species that donate proton known as acid. If the compound…
Q: Determine correct conjugated acid and base pair NO2- +H2O yields HNO2 + OH-
A: Explanation : 1. The acid is a proton donor ions. 2. The base is a proton acceptor ions. 3. We…
Q: 2eq CH.SH dilute Acid HO. 1. CH,M Br 2. H,0 но, н CH, KCN, H-
A: All given reactions are examples of addition reaction on carbonyl
Q: How many acidic hydrogens are present in the molecule shown below? 3 4 1 None
A:
Q: 2.51 Rank the compounds in each group in order of increasing acidity. a. b. C. OH CI CI NH2 OH OH SH…
A: Acidity of a compound means how easily a compound release hydrogen ions. Organic compounds are the…
Q: Answer each question about oxycodone, a narcotic analgesic used for severe paina.Identify the…
A: Since we only answer upto 3 sub-parts, we'll answer the first 3. Please resubmit the question and…
Q: What is the conjugate base to NH4+ ? Select one: a. NH2 - b. NH2- c. NH
A: The congujate base of NH4+ is formed when 1 proton is removed from it.
Q: Which of the following best describes the molecule below? 0000 strong acid neutral weak acid Ostrong…
A:
Q: The pKa of ascorbic acid (vitamin C, page 55) is 4.17, showing that it is slightly more acidic than…
A: Introduction: The acid or base form after the losing or gaining a proton is called conjugate acid…
Q: Which of the following acids is the strongest? A. Ascorbic acid, pK₂ = 11.3 B. Chlorous acid, pK₂ =…
A: Note : Lower the Pka value higher the acidity. Here trifluoro acetic acid pka value = 0.23.…
Q: The pKa of ascorbic acid (vitamin C, page 55) is 4.17, showing that it is slightly more acidic than…
A: Introduction: The acid or base form after the losing or gaining a proton is called conjugate acid…
Q: What is the conjugate base of HSO4°? 22 SO42 H2SO4 S03 H2O II III IV Multiple Choice II II IV
A: Given ion is HSO4- It is an acidic anion because it donates H+ in aqueous solution.
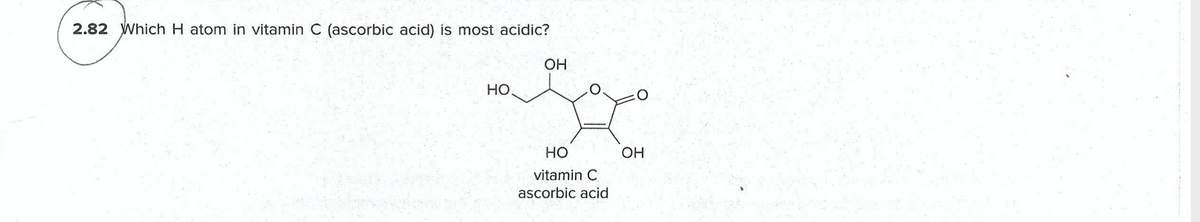
Q: 2.82 Which H atom in vitamin C (ascorbic acid) is most acidic? OH но. но vitamin C ascorbic acid
A:
Q: a) Which of the two molecules is more acidic and why? Please circle your choice R =H R b) Which of…
A:
Q: 4. Circle the list that is ranked from most basic to least basic. H2O NH3 NaH NH3 NaH CH4 a) b) c)…
A: Of the given compounds, the basic strength is decided on the ability of the central atom to donate…
Q: For each structure on the left, pick an identification on the right. Clear All но- -(CH2)4 -OH amino…
A:
Q: Compound name ph-C (CH3)2-0-0-3 (СH3)С-ph PҺCH-CH2 AIBN II || СH3-ҫ-о-о-с-CHЗ ph-c-o-o-ċ-ph ||
A: The first compound name is Bis(2-phenyl-2-propyl)peroxide IUPAC name of this compound is…
Q: a) Ph b) Ph. P2S5 S-Buli -78 °C, THF Ph C) Ph. d) heat 1) NABH4 2) H30* -CO2
A:
Q: Given the following Ka values, which anion is the strongest base? (LOOK AT PICTURE) A. HCO3-…
A: The given values of Ka:
Q: The of formic acid is pKa 3.750. What is the Ka of formic acid?
A: The relationship between Ka and pKa is pKa = -log(Ka ) => Ka = 10-pKa
Q: Ithough you may not think of these compounds as being acids, your organic chemistry instructor…
A: Correct answer for the following question is:
Q: Below there are two groups of compounds. In each group, circle the most acidic compound. 1.c.i. 'N'…
A:
Q: 7. Draw a circle around the most acidic molecule in each pair. OH OH OH OH Vs Vs NC Но ČN Vs
A: Acid is substance which release hydrogen ions when dissolved in water.
Q: Which of the following is the most basic molecule? Selected Answer: Answers: fz6 NH₂ OH NH₂ OH
A: Basic molecules are those that can either accept a proton, H+ (Bronsted-Lowry base) or donate a lone…
Q: 3) Aşağıdaki alkolü fenilasetaldehit ve uygun Grignard reaktifi ile sentezleyiniz. Reaksiyon…
A: The structure of the phenylacetaldehyde is as follows: And the given synthesis is as follows:
Q: which og the following functional groups is basic? a. corn syrup b. melted butter c. coffee d. h2o…
A: The functional groups present in the following compounds are, Corn Syrup: It is also known as…
Q: Which is the weakest base below? O CI-1 O OH-1 O F-1 O NH2-1
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- In the pic (yellow circle l). What is the above O means? Is it 5OH^- or 5 O^- H?naoh mol is 0.11Answer each question about oxycodone, a narcotic analgesic used for severe paina.Identify the functional groups in oxycodone. b.Classify any alcohol, amide, or amine as 1°, 2°, or 3°. c.Which proton is most acidic? d. Which site is most basic? e.What is the hybridization of the N atom? f. How many sp2 hybridized C atoms does oxycodone contain?
- Compared my ao values for NH4Cl=4.143 angstrom and ao values for NaCl = 5.996 angstrom to the literature values ao of NH4Cl = 3.88 Angstrom and ao = 5.64 angstrom. Can you please explain any reason for this including uncertainties.need some help number 3 letters a, b, cThe Ka1 of Oxalic acid is 0.056 The Ka2 of HC2O4- is 5.1 • 10^-5
- USE THE CONDENSED STRUCTURAL FORMULA IN WRITING THE ORGANIC COMPOUND AND GIVE THE NECESSARY COEFFICIENT BESIDE EACH COMPOUND IF NEEDED ANSWER ONLY 7 AND 8the context is "Cosmone is a molecule used by fragrance manufacturers to provide a rich and elegant musky essence to many perfumes. Cosmone has the molecular formula C15H26O." I need help on part (e) and (f).What are the coefficients of NH3 and H2O?___ Al + ___ NO3- + ___ OH- + ___ H2O→ ___ Al(OH)4- + ___ NH3 Group of answer choices 3, 12 4, 18 2,12 3, 14 3, 18

