Atomic Structure
The basic structure of an atom is defined as the component-level of atomic structure of an atom. Precisely speaking an atom consists of three major subatomic particles which are protons, neutrons, and electrons. Many theories have been stated for explaining the structure of an atom.
Shape of the D Orbital
Shapes of orbitals are an approximate representation of boundaries in space for finding electrons occupied in that respective orbital. D orbitals are known to have a clover leaf shape or dumbbell inside where electrons can be found.
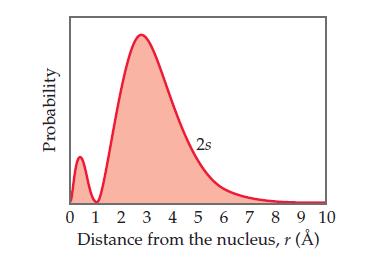
The radial probability function for a 2s orbital is shown here.
Classify the following statements as either true or false: (a) There
are two maxima in this function because one electron spends
most of its time at an approximate distance of 0.5 Å from the
nucleus and the other electron spends most of its time at an
approximate distance of 3 Å from the nucleus. (b) The radial
probability function shown here and the probability density
[ψ(r)]2 both go to zero at the same distance from the nucleus,
approximately 1 Å. (c) For an s orbital, the number of radial
nodes is equal to the principal quantum number, n.
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images









