Q: Which components provide anti-bleeding activity?
A: Heparin is used to prevent or treat certain blood vessel, heart, and lung conditions. Heparin is…
Q: phorus en
A: Octet rule says that elements tend to bond in such a way that each atom has eight electrons in its…
Q: Question 1: In the early 1960s, retroviruses were determined to cause many avian cancers. As a…
A:
Q: A mixture of butene, C4Hs, and butane, C4H10, is burned in air to give CO₂ and water. Suppose you…
A: Numbers of moles is calculated by dividing the given mass with molecular mass.
Q: 2CH3CHO + 5O2 -----------> 4CO2 + 4H2O In the equation above, how many molecules of O2 are required…
A: Given that, 2CH3CHO + 5O2 -----------> 4CO2 + 4H2O then,
Q: The following structures are OH O identical O diastereomers NHCH3 Oenantiomers O constitutional…
A: We have to select the correct option for the given two structures
Q: If you dissolve 2.00 mol KI in 1.00L of water, why will the solution you made not have a molarity of…
A: Molarity is defined as the moles of solute present per litre of solution.
Q: Find the products using HI in the reactions
A:
Q: Tylenol is a widely used over-the-counter analgesic and antipyretic . The molar mass of Tylenol is…
A:
Q: III. Compare the pKa of acetic acid in water, carbon tetrachloride, and dimethyl sulfoxide (these…
A:
Q: Estimate the IR stretching frequency (in cm¹) for the indicated bond in the molecule below. Do not…
A:
Q: Which compound of Grp 3 cations is a white, gelatinous precipitate that produces a colored…
A:
Q: At a certain temperature this reaction follows second-order kinetics with a rate constant of 37.3M…
A:
Q: Quien below Some ontions & which of the tectrolyes NHued, NH₂OH, CH3Cook, weak H₂B03, CarOHT₂, HNO3,…
A: Electrolytes : These are chemical species which conduct electricity when dissolved in…
Q: Unknown X mixed in a 50/50 ratio with acetaminophen yielded a melting range of 154-162°C · -…
A:
Q: B. MnS C. Mg(OH)2 D. FeCO3 Complete the following statements by entering the letter(s) corresponding…
A:
Q: What is the common name for 1,3-dimethylbenzene? A) ortho-xylene B) cumene C) para-xylene Question…
A:
Q: Determine the percent yield for the reaction between 3.2g of Na and excess O2 if 4.1g of Na2O is…
A:
Q: 9.562×10-4 mol of SF6(g) (sulfur hexafluoride gas) is generated into a 192.0 mL headspace, at 25.0…
A: The molar volume of the gas can be calculated by the ideal gas equation which is PV=nRT, where P, n,…
Q: What is the theoretical yield and limiting reagent for the reaction of benzophenone with sodium…
A:
Q: Some HI is placed in a sealed flask and heated to 793 K. When equilibrium is reached, the flask is…
A:
Q: 5, 11.4 & 11.7) Draw the Lewis structure of formaldehyde, CH₂O and then identify electron geometry,…
A: Hybridization is defined as intermixing of orbitals having different energy so as to rehybridize to…
Q: 2. Use Ring A in part a to answer parts a-c. Use a molecular model kit to assist you. a. Complete…
A: Gauche Interaction- The relationship between two atoms or group whose dihedral angke is more than 00…
Q: 2NaHCO3 \rightarrow→ Na2CO3 + CO2 + H2O Which type of chemical reaction does this chemical…
A:
Q: QUESTION 27 Which of the following is the most stable radical? L CH₂=CH IL CH=CHCHCH V. ш сненсн IV.…
A: Radicals are electron deficient species with a carbon having an unbounded electron.
Q: ●● Draw the Lewis structure of NH,OH and then choose the appropriate pair of molecular geometries of…
A:
Q: If the pH = 9, which is true? Question 12 options: [H3O+] is 1 x 10-9M and higher than [OH-]…
A:
Q: Molecular Lewis Formula Structure 1. CIF3 2. BC13 3. CC14 Total Nonbonding Electron Pair Geometry…
A: We are only allowed to solve 1st three subparts acc. to guideline. Thank you.
Q: From the information given below, calculate the heat of combustion of propane (C3H₂) in kJ/mol. C3H8…
A:
Q: (12.8)A substance is initially at 301 K and 1.01 bar (pressure unit). What phase transition occurs…
A:
Q: You have 250. mL of 0.125 M HCl. Using a volumetric pipet, you take 50.00 mL of that solution and…
A:
Q: H3C- ~ OH H H :ÖH H3C-C-H H Aco Dess-Martin periodinane provides a means to oxidize a primary…
A: In a reaction mechanism, the curved arrows describe the movement of electrons. The movement of…
Q: The density of aluminum is 2.7 g/mL. How many liters would 12.8 pounds of aluminum take up?
A: Given, Density of aluminium = 2.7 g/mL Note: 1 pound = 453.592 g Mass of aluminium = 12.8 pounds =…
Q: Aldonic or uronic? НО Uronic acid Ketose OH OH Aldonic acid Aldose Опи OH O OH
A: Aldonic acid : The general formula for the class of compounds is : COOH-(CHOH)n-CH2OH Uronic acid :…
Q: How many Faraday is needed to deposit 5 moles of Magnesium (Mg) from MgF2 solution using…
A:
Q: Molecular Lewis Formula 4. SC12 5. SbCls 6. SF4 Structure Total Nonbonding Electron Pair Geometry…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: (B):-During corrosion tests, current density of 0.86 Amp/m² was measured for steel sample immersed…
A: Given as, Current density, j = I/A = 0.86 Amp/m2 Molecular weight of the steel, M = 55.58 g/mole…
Q: Analyze the H-NMR and C-NMR data below 10 9 8 7 H-NMR 6 200 180 160 140 120 C-NMR ppm 100 80 ppm 3 2…
A:
Q: 4-A differential deration cell,corrosion take place from electrode being thoroughly aerated to…
A:
Q: Draw the mechanism for the following reaction. Provide all resonance structures for the arenium ion…
A: This reaction is known as Electrophilic aromatic substitution and Electrophile is Nitronium ion.
Q: Order the following species with respect to C-C bond length (shortest to longest). I. C2H2 II. C₂H6…
A: In this question, we will arranged all these two compounds in increasing order of C-C bond length.…
Q: (10.5 & 12.3) Which of the following molecules have dipole-dipole forces? More than one answer is…
A: we have to select the molecules that have dipole-dipole forces
Q: Rank the following solutions and compounds from the most conductive to the least conductive. LiCl(s)…
A: LiCl (s) is ionic compound so it does not conduct electricity in solid state. However it is strong…
Q: The order of intermolecular interactions from strongest to weakest is: Charge-charge, dipole-induced…
A: Intermolecular interactions are either attractions or repulsions between two or more molecules.…
Q: A) diethyl ether
A: IUPAC nomenclature is used for naming the organic compounds as recommended by international union of…
Q: Complete these Brønsted-Lowry reactions. reaction 1: HCO3 + H+ = reaction 2: HCO3 + OH =
A: Complete the given Bronsted-Lowry reaction ?
Q: Rank the following solutions from the most conductive to the least conductive. All solutions are 0.1…
A: The conductivity measure the ability of transfer of current in an aqueous solution. The transfer of…
Q: A 2.984 g sample containing impure CuCl₂ 2 H₂O was heated to constant mass. The sample mass after…
A: Given, mass of impure CuCl2.2H2O = 2.984 g Mass sample after heating to drive off the water = 2.452…
Q: For each of the salts on the left, match the salts on the right that can be compared directly, using…
A:
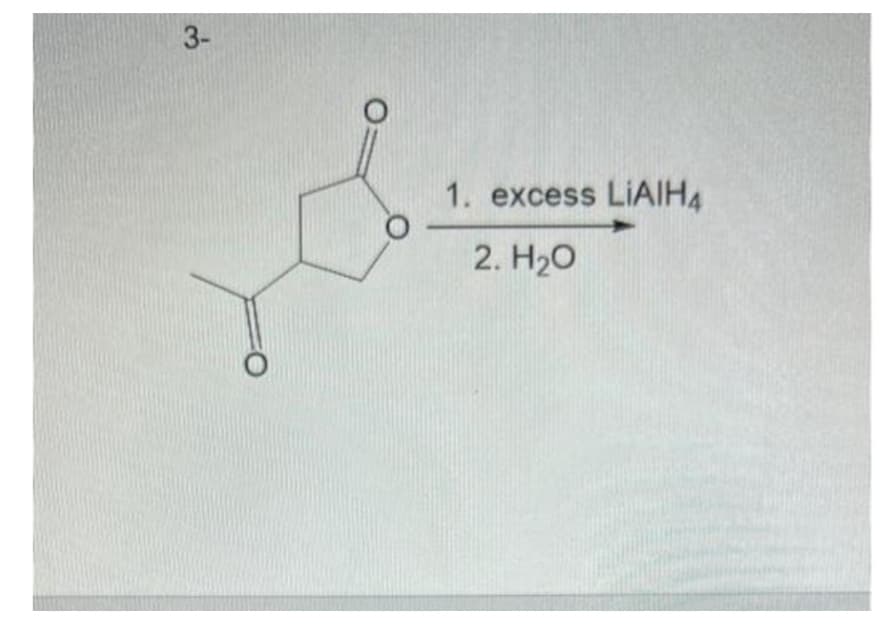
Q: Chemistry Question
A: Valence electrons are the electrons present in the outermost shell or valence shell of an atom. In…
Step by step
Solved in 2 steps with 2 images

- How to manipulate formula mgh=1/2mv2 to find velocity and massActivity 3-2 part1. Answer only no. 1The mercury in a 0.8142-g sample was precipitated with an excess of paraperiodic acid, H5IO6: 5 Hg 2+ + 2 H5IO6 ---> Hg5(IO6)2 (s) + 10 H + The precipitate (MW = 1448.8 g/mol) was filtered, washed free of precipitating agent, dried, and weighed, and 0.4114 g was recovered. Calculate the a) % Hg (200.6 g/mol) b) % Hg2Cl2 (472.1 g/mol)
- To determine the mass of Fe2O3 from the given mass of K3Fe(CN)6, the gravimetric factor to be used is a. Fe2O3 / K3Fe(CN)6 b. Fe2O3 / 2K3Fe(CN)6 c. 2Fe2O3 / K3Fe(CN)6 d. 3Fe2O3 / 2K3Fe(CN)6avg concentration: 8.937x10^-750.00 cm3 of a 1.5784 mol.dm-3 solution of potassium hydroxide is transferred to an empty 700.00 cm3 volumetric flask. This flask is made up to the mark with distilled water and then shaken well. The concentration of the potassium hydroxide in this second flask is:
- A mixture of pure BaCO3 and pure Na2CO3 weighs 1.000 g and has the total neutralizing power of 15.37 meq of CaCO3. Calculate the percentage of combined CO2 in the mixture and the weight of Li2CO3 that has the same neutralizing power as 1.000 g of the above mixture. Note: include up to 4 decimal places Please explain each stepplease answer 1 and 2, thank you!What is the Ksp of Zn3(PO4)2 (MW = 320.8 g/mol) if its solubility in water at 25 °C is 4.9 x 10-5 g/L? format for the scientifc notation 1.0 x 10^-1

