Chapter13: Structure Determination: Nuclear Magnetic Resonance Spectroscopy
Section13.1: Nuclear Magnetic Resonance Spectroscopy
Problem 1P
Related questions
Question
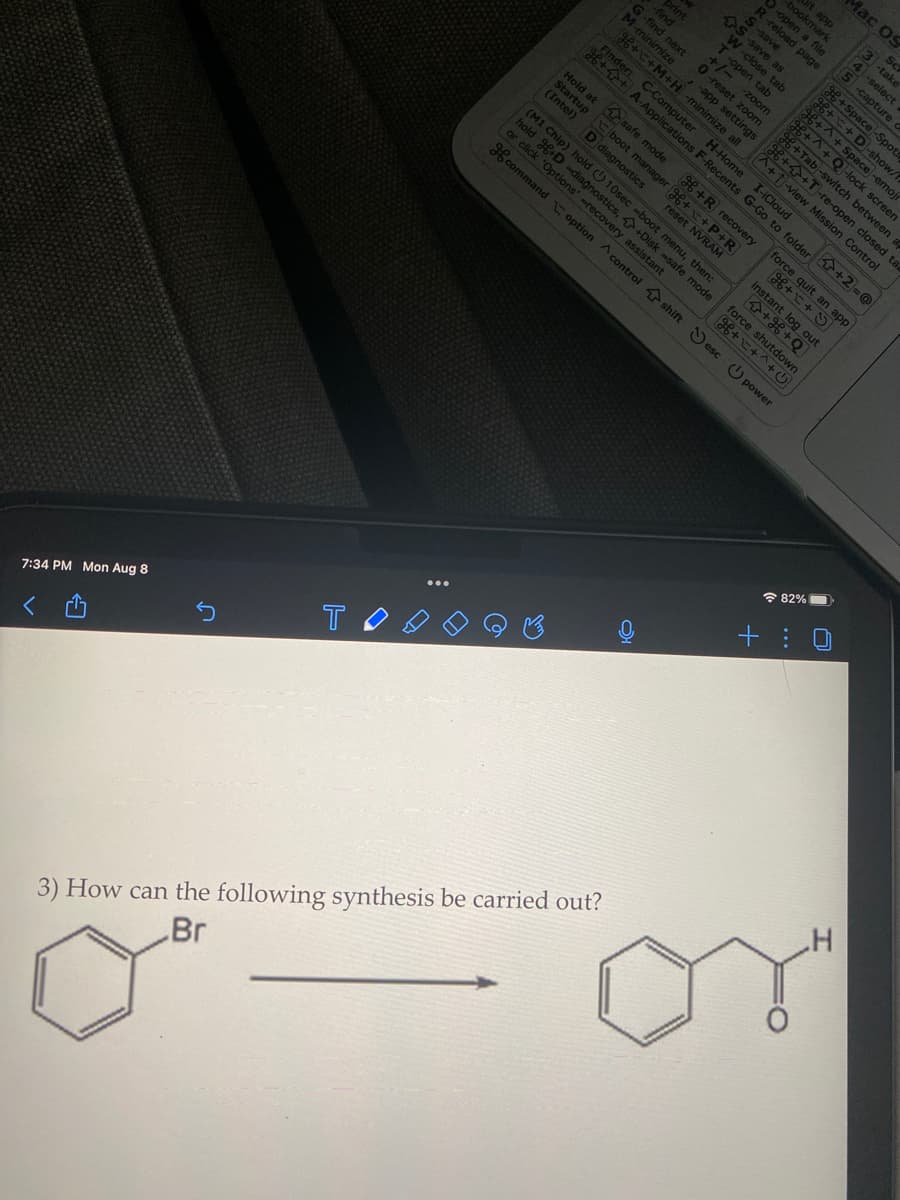
How can the following synthesis be carried out?
Transcribed Image Text:7:34 PM Mon Aug 8
T
Hold at
Startup
(Intel)
-9
3) How can the following synthesis be carried out?
Br
M-minimize
-find
print
-find next
M-minie
+/- -zoo
0-reset zoom
-app settings
-zoom
g++M+H -minimize all
+ C-Computer
D diagnostics
O
safe mode
boot manager g++P+
reset NVR
bookmark
app
as
-close tab
H-Home
^+T-re-open closed ta
+T-view Mission Control
I-iCloud
A-Applications F-Recents G-Go to folder +2=@
force
(M1 Chip) hold 10sec -boot menu, then:
hold +D -diagnostics,
or click 'Options' -recovery assie safe mode
stant
dar+^+
H command option control shift esc power
d nage
+R recovery
Instant
+ app
C+Y
shutdown
out
Mac OS
H+Space-Spotr
g++ Space-emoj
++ D-show/f
g6+ +^+Q-lock screen
+Tab-switch between a
82%
Sc
3-take
4-select
5-capture d
+:0
.H
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning