3. Completely explain the following observation: Br Br2 Br₂ Br Br + HBr A. Both reactions involve two bromine atoms adding across a double bond B. Both reactions involve a neutral intermediate with a single bromine atom C. After addition to benzene, elimination is favourable to regain aromaticity D. After addition to cyclohexene, the addition of a second bromine atom is favourable
3. Completely explain the following observation: Br Br2 Br₂ Br Br + HBr A. Both reactions involve two bromine atoms adding across a double bond B. Both reactions involve a neutral intermediate with a single bromine atom C. After addition to benzene, elimination is favourable to regain aromaticity D. After addition to cyclohexene, the addition of a second bromine atom is favourable
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter21: Benzene And The Concept Of Aromaticity
Section: Chapter Questions
Problem 21.16P: Which of the molecules and ions given in Problem 21.15 are aromatic according to the Hckel criteria?...
Related questions
Question
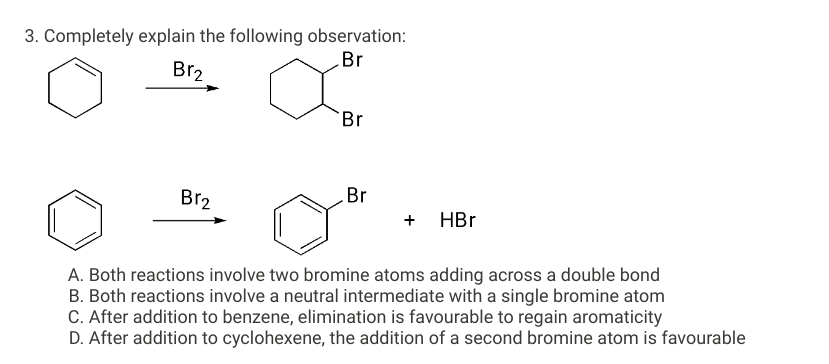
Transcribed Image Text:3. Completely explain the following observation:
Br
Br2
Br₂
Br
Br
+ HBr
A. Both reactions involve two bromine atoms adding across a double bond
B. Both reactions involve a neutral intermediate with a single bromine atom
C. After addition to benzene, elimination is favourable to regain aromaticity
D. After addition to cyclohexene, the addition of a second bromine atom is favourable
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning