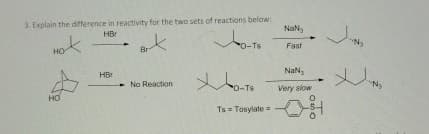
3. Explain the difference in reactivity for the two sets of reactions below: HO HO HBr Br До-то HBr No Reaction Хо-тв Ts=Tosylate NaN, Ny Fast NaN, Very slow ・ N
Q: A solution is prepared that is initially 0.41 M in pyridine (C,H,N), a weak base, and 0.20M in…
A: initial changefinal
Q: Draw all of the stereoisomers of the structure shown below. Use a dash or wedge bond to indicate the…
A: A pair of substances which are non-super imposable mirror images are called enantiomers.A pair of…
Q: 3. Calculate the pH of a 1.94x103 M solution of CH COOH at 25°C. K, 4.55x100
A: Concentration of acetic acid(CH3COOH) = 1.9410-3 MKa of CH3COOH = 4.5510-10
Q: 4) Which of the following statements is true of a catalyst? A) Its concentration stays constant…
A: The catalyst is a chemical species that increases the rate of a chemical reaction and it does not…
Q: ... 1. Draw by hand a sawhorse conformation of the structure below. CH3 0 HO OCH3 NH2
A: Since you have asked multiple question, we will answer the first question for you. If you want any…
Q: 1. Show how you would synthesize each compound from starting materials containing no more than six…
A: In the synthesis of the first compound the reaction of benzene with acid chloride and AlCl3 is…
Q: 5. For the following molecules, draw the fragments based on the peaks listed. Consider the molecular…
A: Mass spectral analysis is used for the structural elucidation of organic compounds. The compound is…
Q: Reduction In organic chemistry, reduction is defined as a reaction in which a carbon atom forms…
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: Which statement about the charge-minimized Lewis structure of azide (N3) is true? (Charges not shown…
A:
Q: Only typed solution
A: The objective of the question is to understand the reasons behind the increase in pH and PO2 in the…
Q: 0,0,. Write the empirical formula for at least four ionic compounds that could be formed from the…
A: The objective of this question is to determine the empirical formulas for ionic compounds that could…
Q: Please write a paragraph of the objective stated using the two images attached below Please please…
A: Acid-base titrations are analytical methods that are employed in the determination of an unknown…
Q: Predict the major product obtained when benzoic acid is treated with fuming sulfuric acid. Draw the…
A: The starting material is given as benzoic acid:This is treated with fuming sulfuric acid.
Q: Identify the missing reagent(s), reactant(s) or final product(s). Some transformations may require…
A: Given are organic synthesis reactions. All reactions involve different types of reactions.
Q: Odify the Fischer projection of the following aldose to show the compound that is formed when the…
A:
Q: 1. Predict the product and provide the mechanism for the following reversible nucleophilic addition.…
A: See answer in the explanation fieldExplanation:
Q: If the reaction quotient is less than the equilibrium constant, which statement would be correct? O…
A: Given that,Reaction quotient Q is less than the equilibrium constant K.Choose the correct option.
Q: Synthesize the following stereoisomers utilizing a strategy that makes the desired isomer a major…
A: In the given question we have to predict the reaction conditions for the formation of the desired…
Q: Part C MISSED THIS? Watch KCV: The Titration of a Weak Acid and a Strong Base, IWE: Weak Acid-Strong…
A: The question is based on the concept of the pH of the solution.It is defined as a negative logarithm…
Q: 1. Calculate the concentration of a solution of H₂SO, with a pH of 1.06.
A: 1) pH = - log [H+] Thus, [H+] = 10-pH
Q: Draw the major product of this reaction. Include stereochemistry if applicable. Use a dash or wedge…
A: It involves an elimination reaction - E2 (bimolecular elimination) reaction. This reaction proceeds…
Q: GIF Add the missing curved arrow notation, lone pair electrons, and non- zero formal charges. NV +…
A: The given reaction is based on nucleophilic substitution reaction.
Q: The fraction of sites bound on the particular macromolecule is 0.53 at a ligand concentration of…
A: Given:Fractional saturation of binding sites = 0.53Concentration of Ligand ([L]) = 0.01992…
Q: . At 20°C, a 0.376 M aqueous solution of ammonium chloride has a density of 1.0045 g/mL. What is the…
A: The concentration of a solution can be represented in different ways. Molarity indicates the amount…
Q: Draw the major product of this reaction. Ignore inorganic byproducts.
A: LDA is a sterically hindered strong base and it is used in the formation of the kinetic enolate ion.
Q: An aluminum cup weighing 60.0 g is filled with 30.0 g of water. How much energy is needed to raise…
A: FINAL ANSWER: How much energy is needed to raise the temperature of the water filed cup from…
Q: Calculate the pH of a 0.21 M solution of barium hydroxide. Express your answer in decimal notation…
A: Concentration of barium hydroxide solution = [Ba(OH)2] = 0.21 MpH of the solution = ?Note: Barium…
Q: What is the missing reagent in the reaction below? Ph Op a Br Ph.P
A: The objective of the question is to predict the reagent used in the given reaction.
Q: 4. Provide a mechanism for the following transformation: NaOH H2N. Н20, д
A:
Q: Mechanism 1. Provide the complete mechanism for the reaction below. You must include appropriate…
A: Diels-Alder reaction is a type of electrocyclic reaction which involves the cycloaddition between…
Q: Draw all of the stereoisomers of 2-chloro-5-methylheptane. Use a dash or wedge bond to indicate the…
A: Stereoisomers are molecules in which the atoms are connected in the same order, but the spatial…
Q: Please don't provide handwriting solutions..
A: Given,The reaction:
Q: 3. Complete the following table: Fill in the mL used of the two starting materials (from the…
A: The answers and calculations are as follows.Explanation:
Q: arrow. F Cl HS HC H SH Part 1 of 2 Draw the Newman projection for a staggered conformation of the…
A: The objective of the question is to find the Newman projection for one staggered and one eclipsed…
Q: 1 & 2. Identify the steps necessary to prepare compound X. Show reagents and reaction conditions.…
A: To approach the product from cyclopent-1-en-1-ylbenzene to…
Q: For each of the following compounds, decide whether the compound's solubility in aqueous solution…
A: Solubility of a compound changes with PH, if any constituent ion of the compound reacts with Proton…
Q: The acid ionization constant for Pb(H2O)62+(aq) is 6.3×107. Calculate the pH of a 0.0651 M solution…
A:
Q: You will dilute ~1.00 g of solid acid in a flask. To what volume in mL will you dilute it? Type…
A: To calculate the volume of solution needed to dilute 1.00 g of solid acid, you can follow these…
Q: Please help me , I need fast solution. Please don't parovide handwritten solution ....
A: The objective of the question is to complete the given chemical reaction involving CH2I2, Zn(Cu) and…
Q: Which of the following is considered to be a thermodynamic product of the reaction shown. DBr OA OB…
A: Diene shows the electrophilic addition reaction such addition of X2, HX etc. The addition of HX to…
Q: Which two of the following compounds yield the same carbocation upon ionization? Check the two that…
A: -> If leaving group removed from molecule then there is formation of carbocation. If possible…
Q: NH3 is a weak base (K₁ = 1.8 x 10-5) and so the salt NH4Cl acts as a weak acid. What is the pH of a…
A: NH4Cl is a salt of weak base NH3 and strong acid HCl. So in an aqueous solution of the salt the salt…
Q: Complete the table below by deciding whether a precipitate forms when aqueous solutions A and B are…
A: Given, Solution A Solution B Does a precipitation forms when A and B…
Q: 1) The product of the reaction shown is which of the following? 1) NaBH4 CI 2) H3O+ a. An aldehyde…
A: C. An AlcoholExplanation:Approach to solving the question:The reaction is the reduction of carbonyl…
Q: For the reaction below, Kc = 0.60 at 550 K. The reaction starts with a 0.050 M concentration of…
A: The objective of the question is to calculate the expression for the product at equilibrium.
Q: Only typed solution
A: The objective of the question is to identify the possible product formed in the vial due to the…
Q: The equilibrium constant for the chemical equation N2(g) + 3H2(g)2NH3(g) is Kp = 0.180 at 213 °C.…
A: Given reaction,The pressure equilibrium constant,The temperature of the gas,The concentration…
Q: Sulfur dioxide and oxygen react to form sulfur trioxide, like this: 250(g)+0₂(g) 250 (9) The…
A:
Q: aming and 1. Draw and name the following compounds ✓a. CH CHCHCHICH
A: Given are condensed structural formulas for organic compounds. Rules for determination of IUPAC…
Q: 5. A solution of HF is found to be 0.25M and have K, value of 6.6x104. a. Write the equilibrium…
A:
Unlock instant AI solutions
Tap the button
to generate a solution
Click the button to generate
a solution
- Which compound is not a possible product in the reaction below ( F2 is in excess F2and UV light the catalyst).CHA + F2 -->O CFAO CHFaO CHF-O CH2F2O CHaFConsider the reactions shown below. For each reaction state if it would favor S N 1 or S N 2pathway and give the major product of the reaction.Arrange the alkyl halides in order of increasing reactivity in an SN2 reaction with KI in acetone (least first). I, IV, III, II II, III, I, IV IV, I, III, II III, II, IV, I
- [IV1] Instructions: Follow the flow of electrons indicated by the curved arrows in each of the following reactions, and predict the products that result: (refer to the photo below)Identify the reagents needed to come up with the given compounds. Pls Separate the reagents with a comma and a space. (ex. HNO3, H2SO4). For a multi-step reaction, separate the reagents with a semi-colon. (ex. HNO3, H2SO4; CH3Cl, AlCl3) There should be 9 reagents1) The Predict the product(s). 2)Label any kinetic/thermodynamic product(s). 3)Give reason towards the selection of kinetic product(s).
- 6. For each of the following reactions: a) say what is the expected Mechanism sn1 or sn2 and say why. b) For some of the reactions say if the product is optically active and say why. c) For some of the rxns say if their kinetics are unimolecular (Uni) or bimolecular (Bi) and explain MechanisOrgonic Chmeistry II: 2,4,6-Trinitrotoluene (TNT) is synthesized by trinitrating toluene. The first nitration proceeds much faster than the second two. Briefly explain.Arrange the nucleophiles in order of increasing reactivity (least first). III, II, I, IV IV, III, I, II IV, I, II, III III, I, IV, II
- Why we need step 3 before step 4? a. Because the nitro group increases the electrophilicity at the ortho positions which is where the bromine is added. b. Because the amine group is a strong ortho, para director which is what controls the regiochemical outcome of this bromination. c. Step 4 is unessesary. The symmetry of compound 3 allows for the bromination to be regioselective and give compound 5. 5. There will be a mixture of products because there is no selectivity for a major product.Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) Br2, FeBr3 Na/NH3, -33 degrees C NBS, light KMnO4, H3O+ Mg metal, ether KOH, EtOH, heat(b) Predict the suitable solvent (H2O or CH3COCH3) to increase the reaction of bromopropane(CH3CH2CH2Br) with sodium hydroxide (NaOH). Two reactions are shown below:

