3. SEP Analyze Data Calculate the ratio of the moles of product produced to the moles of each of the reactants used. (Write two separate ratios.)
3. SEP Analyze Data Calculate the ratio of the moles of product produced to the moles of each of the reactants used. (Write two separate ratios.)
Chapter11: Solving Equilibrium Problems For Complex Systems
Section: Chapter Questions
Problem 11.3QAP
Related questions
Question
100%
hello please help to answer #3 using the data shown below
Transcribed Image Text:ships in Chem. Rxns (17-E1) ☆
-ons Help Last edit was 3 minutes ago
Arial
-
12 +
M
B I U A
E- E ===
1
2 |
3
11
I
4
** 5 11 [
1
6
3. SEP Analyze Data Calculate the ratio of the moles of product produced to the
moles of each of the reactants used. (Write two separate ratios.)
4. SEP Interpret Data Derive the stoichiometric ratios for this chemical equation (in
other words, balance the equation using the data from the experiment). Do the
toichiometric ratins match what you would expect from balancing the equation
GO
+
A
13
TURN
Transcribed Image Text:produc
0
moles
do 16
9. 14
0.12
0.1
४
9.6
0,4
0.2
0
2
0
0.15 0.2 0.25
moles of reactant
(12/unligning
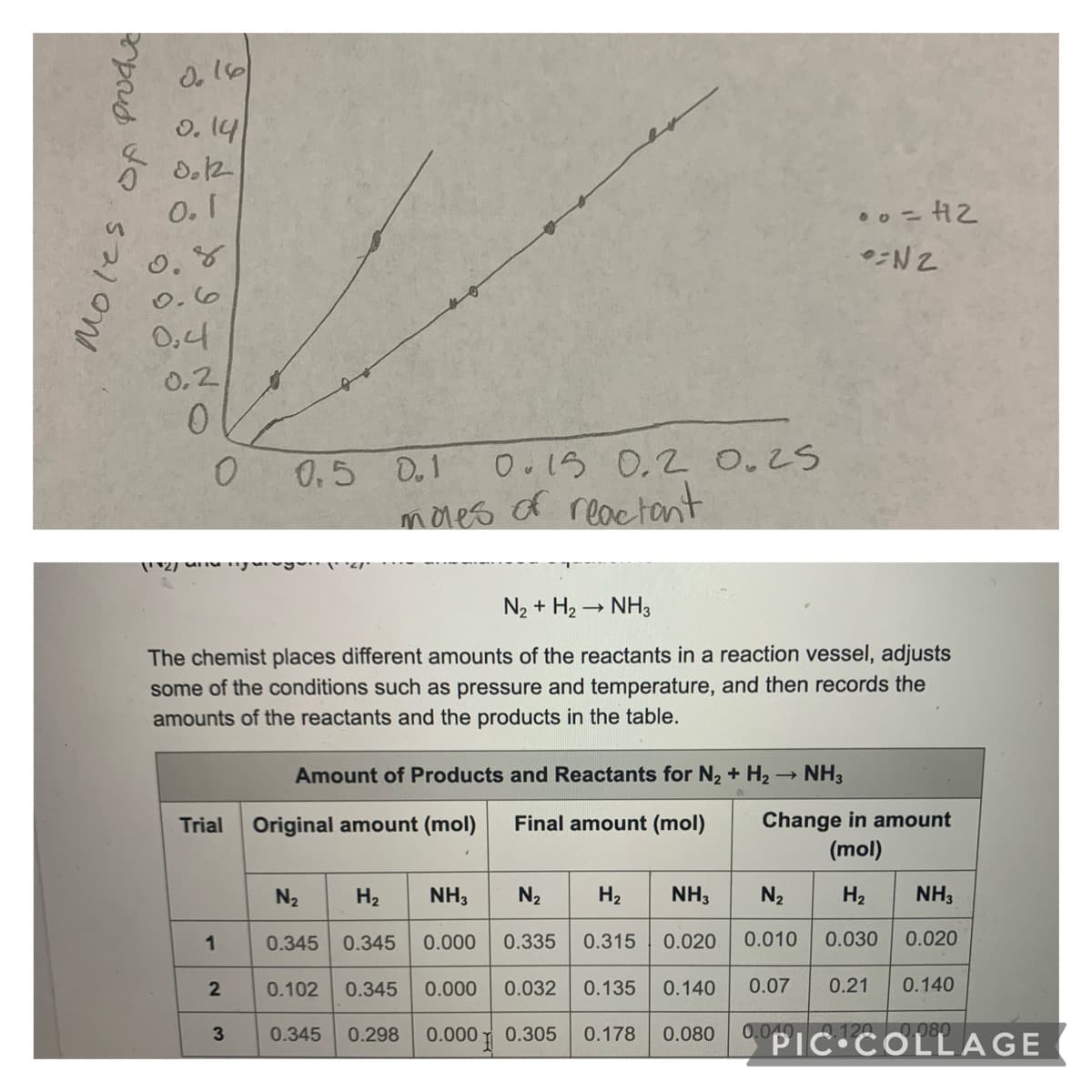
N₂ + H₂ → NH3
The chemist places different amounts of the reactants in a reaction vessel, adjusts
some of the conditions such as pressure and temperature, and then records the
amounts of the reactants and the products in the table.
Amount of Products and Reactants for N₂ + H₂ → NH3
Trial
Original amount (mol)
Final amount (mol)
Change in amount
(mol)
N₂
H₂
NH3
N₂
H₂ NH3
N₂
H₂
NH3
1
0.345 0.345 0.000 0.335
0.315
0.020
0.010
0.030
0.020
0.102 0.345
0.000 0.032
0.135 0.140
0.07
0.21 0.140
0.345 0.298
0.000
0.305
0.178 0.080
0.040 0.120 0.080
3
0 = H2
0.5 0.1
00-
•N2
PIC COLLAGE
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning