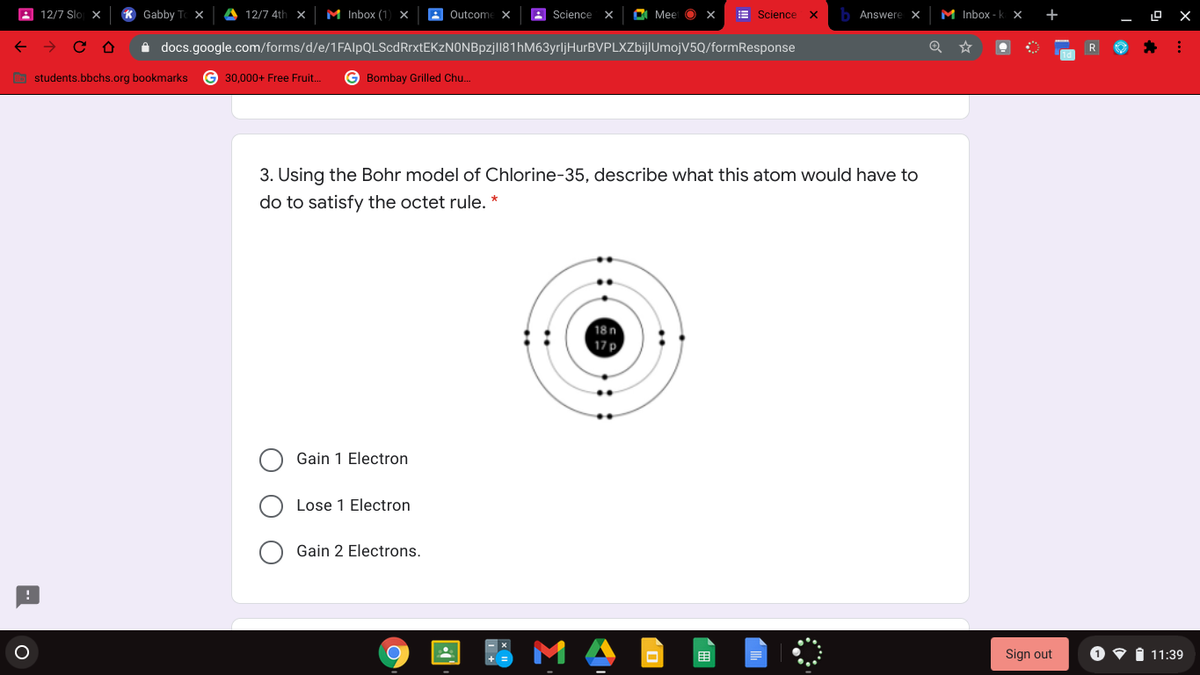
3. Using the Bohr model of Chlorine-35, describe what this atom would have to do to satisfy the octet rule. * 18n 17p Gain 1 Electron Lose 1 Electron Gain 2 Electrons.
3. Using the Bohr model of Chlorine-35, describe what this atom would have to do to satisfy the octet rule. * 18n 17p Gain 1 Electron Lose 1 Electron Gain 2 Electrons.
Chapter5: Errors In Chemical Analyses
Section: Chapter Questions
Problem 5.12QAP
Related questions
Concept explainers
Atomic Structure
The basic structure of an atom is defined as the component-level of atomic structure of an atom. Precisely speaking an atom consists of three major subatomic particles which are protons, neutrons, and electrons. Many theories have been stated for explaining the structure of an atom.
Shape of the D Orbital
Shapes of orbitals are an approximate representation of boundaries in space for finding electrons occupied in that respective orbital. D orbitals are known to have a clover leaf shape or dumbbell inside where electrons can be found.
Question
Using the Bohr modle of chlorine-35, describe what this atom would have to do to satisfy the octet rule.
Transcribed Image Text:A 12/7 Slo x
K Gabby To ×
A 12/7 4th X
M Inbox (1) x
8 Outcome X
Science
O Mee O
E Science
Answere X
M Inbox - k x
+
i docs.google.com/forms/d/e/1FAlpQLScdRrxtEKzN0NBpzjll81hM63yrljHurBVPLXZbijlUmojV5Q/formResponse
O students,bbchs.org bookmarks
G 30,000+ Free Fruit.
Bombay Grilled Chu..
3. Using the Bohr model of Chlorine-35, describe what this atom would have to
do to satisfy the octet rule. *
18n
17 p
Gain 1 Electron
Lose 1 Electron
Gain 2 Electrons.
M A
1 11:39
目
Sign out
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



