Q: The diagram below represents the layout of a typical single-beam UV-Vis, explain the function of eac...
A:
Q: Y CH3CH2CHCH=CH2 + HBr CH3 Major Minor (a) Draw the structure ofY and Z (b) Write the mechanism for ...
A:
Q: Molecule ЕPG Нyb (circle if polar) 2D Lewis Structure 3D Lewis Structure MG ВА AsCl5 03
A:
Q: Develop a general equation for the standard heat of reaction as a function of temperature for one of...
A: ANSWER : (a) C2H2(g) + H2O (g)→(CH2)2O (g)Heat of reaction is as follows ;∆Hrxn= ∑products,ini∆Hf29...
Q: How much HNO3 can be formed in the following reaction if 3.8 kg of NO2 gas are bubbled through 1.0 k...
A: Mass of NO2 = 3.8 kg Mass of H2O = 1.0 kg Mass of HNO3 = ?
Q: The iodination of salicylamide was carried out analogous to Experiment 12 in your “Green Organic Che...
A: Increase in chemical shifts suggest that the proton is deshielded. Hydrogen bond interaction is one ...
Q: Experimental data shows the water-alcohol proportion at 60°C Mole fraction of 0.00 0.05 0.27 0.44 0....
A:
Q: A solution for disinfecting clinical thermometers was prepared by dissolving 22.5 g of ethyl alcohol...
A:
Q: The stepwise stability constant, K at 25 °C in an aqueous solution for the formation of copper and n...
A: stepwise stability constant
Q: II. Analysis. Given below is a schematic diagram for a simple analysis of a novel tetrasaccharide is...
A: D- fructose. B- glucose . C - galactose . As A gives Benedict's test: (+) Seliwanoff's test: (-) Bi...
Q: To get electrical energy from an Galvanic cell the emf should ans. Positive None Zero Negative
A:
Q: A 5.0 L balloon has a pressure of 1 atm at sea level, where the temperature is 28 ͦC. The balloon w...
A: Initial volume = 5.0 L Initial pressure = 1 atm Initial temperature = 28 °C New temperature = 11 ...
Q: 10.00 g NaCl is dissolved in 90.00 mL of water. Calculate the percent by mass-volume of the solution...
A: Given mass of NaCl = 10.00 g Volume of the solution = 90.00 mL. The %(w/v) is
Q: How many moles are there in 7.14 × 10³⁵ molecules of O2 Use the format for solution • Given • Unknow...
A:
Q: Туре of Crystal Covalent Metallic Ionic Molecular Substance Network Dispersion Forces Dipole- Dipole...
A: In this question we have to determine the type of crystal solid formed by the given compounds.
Q: The rate constant k for a certain reaction is measured at two different temperatures: temperature k ...
A: Given data: The initial rate constant = 3.6 ×109 The final rate constant = 1.3 × 109 The initial te...
Q: Which of the following aqueous solutions would NOT form a precipitate when added to a solution of si...
A: When silver nitrate is react with different salt of sodium to produce insoluble compound and a solub...
Q: Under certain conditions the rate of this reaction is zero order in ammonia with a rate constant of ...
A:
Q: O. An acrobat swings from a trapeze with a frequency of 0.5 Hz. What is the length of the trapeze ca...
A: Given : Frequency = 0.5Hz
Q: Naturally occurring Europium (Eu) consists of two isotopes with a mass of 151amu and 153amu. If the ...
A:
Q: 1. Plot (and cite examples) the titration curve of the following: a. strong acid vs strong base b. s...
A: Since you have asked multiple question, we will solve the first question for you. If you want any sp...
Q: When the concentration of H+ ions in a solution decreases, a buffer maintains the pH of a solution b...
A:
Q: 3.30 Name the following cycloalkenes: (a) CH3 (b) (c) (d)
A:
Q: LOME ,个 OMe ÇN
A:
Q: uation: Al,0,(s) + 6NAOH(1) + 12HF(g) 2Na,AlF, + 9H,O(g) 12.6 kg of Al,0,(s), 57.4 kg of NaOH(1), an...
A:
Q: Devise a synthesis of the following compounds starting from benzene.
A: The compound to be synthesized is 5-Bromo-2-nitroaniline. Step 1 Treating benzene with nitrating mix...
Q: Table 3 Temperature C Time in seconds Rate [2/time Value of k !! 46.5 26sec (Using the values of the...
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
Q: Molecule ЕPG Hyb (circle if polar) 2D Lewis Structure 3D Lewis Structure MG ВА CIF*
A:
Q: The relative atomic mass of antimony is 121,8. Antimony exists as two isotopes - antimony 121 and an...
A: Given, The relative atomic mass of antimony is 121.8 and it's two isotopes antimony 121 and antimon...
Q: What is matter
A: What is matter ?
Q: 2. Predict whether the following pairs of liquids would be miscible or immiscible: a. Water and meth...
A: To answer this question, it is very important to consider the polarity index of the mentioned solven...
Q: Which of the following best describes what occurs at absolute zero?
A: Absolute zero is 0K.
Q: Which is true regarding the compound C12H22011 (lactose)? O it is ionic and held together by opposit...
A: A covalent or molecular bond is formed with the sharing of electrons.
Q: QUESTION 14 What is the geometry relative to the O atom in the Lewis structure below? H. H. H-C- C-H...
A:
Q: what is 0.939g of 4-nitrobenzoyl chloride in mmol?
A:
Q: L) One type of commercial smoke detector contains a minute amount of radioactive americium-241 (24'A...
A: Given that
Q: Which reagent(s) are best used to carry out the following reaction? OH a. H2CrO7 b. 1. propanol, NaO...
A: In the given reaction the aldehyde is converted into alcohol by the addition of an alkyl group to th...
Q: you'd like to weigh out acetic acid and sodium acetate (pka=4.75) to use as buffer system to study f...
A: This would simply require the Henderson Hasslebech reaction, which states that when the pKa value of...
Q: What is commonly determined using Atomic Spectroscopy? Bodn energy Elemental composition Concentrati...
A:
Q: A gaseous product has a mass of 2.34g and occupies a volume of 0.854 L.The temperature in the labora...
A:
Q: Synthesize 2,3-Dimethylbutane by Wurtz reaction. Explain each step and draw the mechanism.
A: Wurtz reaction can be defined as the reaction in which alkyl halides react with sodium in the presen...
Q: statement
A: When a pure substance changes phase from subcooled liquid to superheated steam, this CAN be reversed...
Q: A sealed reaction vessel initially contains 1.839×10-2 moles of water vapor and 1.301x10-2 moles of ...
A:
Q: What is the molar mass of a sugar, if a solution of 1.4 g of the sugar in 0.20 L of solution has an ...
A: Please find your solution below : Osmotic pressure is a pressure that should be applied on the solut...
Q: Convert the following into moles if they have 25.5 g of the ff: a. Zinc, b. CuCl2 c. KSO4
A: The relation between mass and number of moles of any substance is given by tge relation - Mole = mas...
Q: Ethyl chloride, CH3CH2Cl, used to produce tetraethyllead gasoline additive, decomposes when heated t...
A: The given reaction is : CH3CH2Cl(g) → C2H4(g) + HCl(g) The reaction is first order. The initial co...
Q: Answer the following. Choose from the provided choices. 1. A molecule has a molecular formula of A2...
A: Here we have to determine the number of valence electrons in X atom in chemical species A2X3^2- in t...
Q: 2 CuCl2 + 4 KI → 2 CuI + 4 KCl + I2 When
A: Given reaction is 2 CuCl2 + 4 KI --> ...
Q: Consider the following reaction where K, = 0.497 at 500 K. PCI5(g) PCI3(g) + Cl2(g) If the three gas...
A: The equilibrium reaction given is, => PCl5 (g) ↔ PCl3 (g) + Cl2 (g) Given: Kp = 0.497 And initia...
Q: Using the figure below, which points show an equilibrium? 1 atm liquid phase soid phase gas phase AT...
A: At the equilibrium point, the rate of the forward abd backward reaction will be equal.
Can you help me answer and explain 3.26
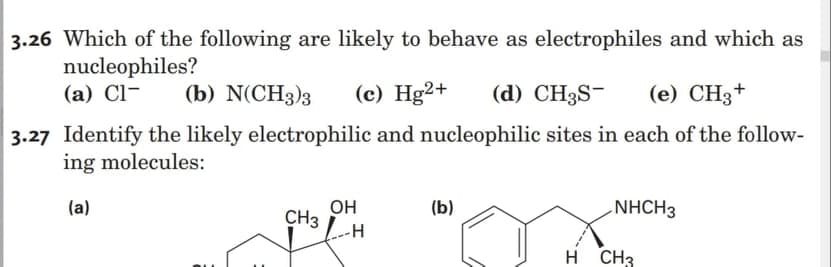
Step by step
Solved in 2 steps with 1 images

- Compound 1 is an anticoagulant that is extracted from, among other plants, the sweetclover plant, Melilotus Officinalis. However, the compound is moderately toxic and alsofinds use as a rodenticide. Name this compoundIn the following three compounds(1,2,3) arrange their relative reactivity towards the reagent CH3Cl / AlCl3. and justify it.What is going on from compound B to compound C? Can you please select all the options that apply
- The hydrocarbon fluorene was treated with potassium t-butoxide in an acid-base reaction, giving the fluorenide anion and t-butyl alcohol. (a) Which way does the equilibrium lie, and by how much? b) What is the proportion of the fluorenide anion to fluorene? (c) Why is fluorene so highly acidic, considering the pKa of an average alkane is above 50?When the nitrogen-containing aromatic heterocyclic compounds 1 and 2 are treated with HCl, only 1 forms the hydrochloride salt, whereas compound 2 is unreactive. Provide an explanation for this observed reactivity.An unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent ofH2 over a palladium catalyst. Hydrocarbon A also reacts with OsO4 to give diol B. When oxidized with KMnO4 in acidic solution, A gives two fragments. One fragmentis propanoic acid, CH3CH2CO2H, and the other fragment is ketone C. What are thestructures of A, B, and C? Write all reactions and show your reasoning.
- Rank each of the following sets of nitrogen bases in terms of basicity and explain your answerCompound K, L and M are three isomers with the molecular formula C5H10O.Compound K cannot be oxidized, while compound M and L can be oxidized.Oxidation of compound L with hot acidified potassium permanganate,KMnO4 solution yields 2-methylbutanoic acid. When treated with Iodoformreagent, yellow precipitation only occurs in compound K. Fehling test onlyyielded positive results for compound L and negative for compound M andK. Compound M is then reacted with hydrogen chloride, HCl produceschlorocyclopentaneCompound K, L and M are three isomers with the molecular formula C5H10O.Compound K cannot be oxidized, while compound M and L can be oxidized.Oxidation of compound L with hot acidified potassium permanganate,KMnO4 solution yields 2-methylbutanoic acid. When treated with Iodoformreagent, yellow precipitation only occurs in compound K. Fehling test onlyyielded positive results for compound L and negative for compound M andK. Compound M is then reacted with hydrogen chloride, HCl produceschlorocyclopentane (i) Draw the structural formula of compounds K, L and M.
- Compound K, L and M are three isomers with the molecular formula C5H10O.Compound K cannot be oxidized, while compound M and L can be oxidized.Oxidation of compound L with hot acidified potassium permanganate,KMnO4 solution yields 2-methylbutanoic acid. When treated with Iodoformreagent, yellow precipitation only occurs in compound K. Fehling test onlyyielded positive results for compound L and negative for compound M andK. Compound M is then reacted with hydrogen chloride, HCl produceschlorocyclopentane. (ii) Name the type of chemical reaction of compound M when reacted withhydrogen chloride, HCl .Two moles of organic compound ‘A’ on treatment with a strong base gives two compounds ‘B’ and ‘C’. Compound ‘B’ on dehydrogenation with Cu gives ‘A’ while acidification of ‘C’ yields carboxylic acid ‘D’ with molecular formula of CH2O2. Identify the compounds A, B, C and D and write all chemical reactions involved.The crocodile, which can remain under water without breathing for up to 1 h, drowns its air-breathing prey and then dines at its leisure. An adaptation that aids the crocodile in doing so it that it can utilize virtually 100% of the O2in its blood whereas humans, for example, can extract only ~65% of the O2in their blood. Crocodile Hb does not bind BPG. However, crocodile deoxyHb preferentially binds HCO3ꟷ. How does this help crocodile obtain its dinner? Explain your answer.

