32. Balance the following equation occurring in an acidic aqueous solution. Indicate which method you have chosen and show all steps relating to the balancing method. Once completed, show one method to check your work. Fe2+ + Cr,0, (aq) Fe3+ (aq) + Cr3+ (aq) (aq) >
32. Balance the following equation occurring in an acidic aqueous solution. Indicate which method you have chosen and show all steps relating to the balancing method. Once completed, show one method to check your work. Fe2+ + Cr,0, (aq) Fe3+ (aq) + Cr3+ (aq) (aq) >
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter3: Chemical Reactions
Section: Chapter Questions
Problem 63PS: Balance each of the following equations, and classify them as precipitation, acid-base, gas-forming,...
Related questions
Question
DO BOTH OF THE QUESTIONS PLS I BEG I SWEAR DO BOTH FUCKING QUESTIONS
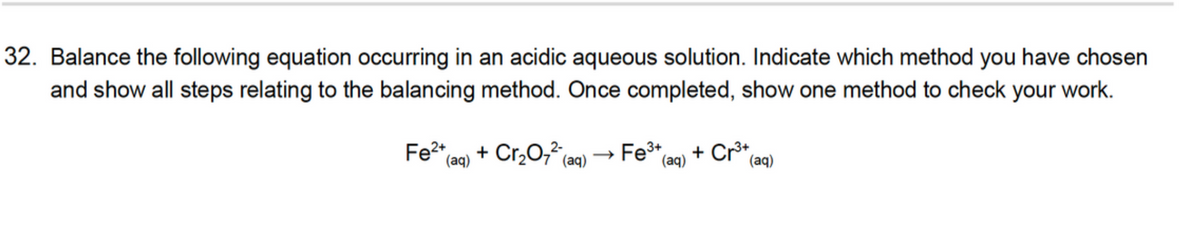
Transcribed Image Text:32. Balance the following equation occurring in an acidic aqueous solution. Indicate which method you have chosen
and show all steps relating to the balancing method. Once completed, show one method to check your work.
Fe2*
+ Cr,0, (aq)
Fe* (aq)
+ Cr*
(aq)
(aq)

Transcribed Image Text:26. (a) What is the process of titration? (b) How does it work? (c) What is at least one practical application
of titration? Explain your ideas in at least three (3) sentences. Be sure to use relevant vocabulary terms.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning