Q: Br 1. PPH3 2. n-butyllithium H. 3.
A: Wittig reaction is the reaction which is used to produce alkene by the help of carbonyl compound. Tr...
Q: n the presence of excess thiocyanate ion, SCN-, the following reaction is first order in iron(III) ...
A:
Q: 4NH3 (g) + 502 (2) 4NO(R) 6H20 (g) --> Answer each question that follows. O a. Increasing the pressu...
A: Given : We have to pick the correct statement for the given reaction.
Q: Suppose a 250. mL flask is filled with 0.50 mol of Br,, 0.10 mol of BROCI and 0.40 mol of BrCl. The ...
A: Calculate the equilibrium molarity of OCl2 ?
Q: Which type of hydrocarbon is represented by this structure? H. H. H. H. Select the correct answer be...
A: ANS. aromatic hydrocarbon
Q: Nitrogen dioxide decomposes according to the reaction: 2 NO2(g) → 2 NO(g) + O2(g); Kp = 4.48 x 10-13...
A: The equilibrium constant of a chemical reaction is the value of its reaction quotient at chemical eq...
Q: Nitrogen and hydrogen react according to the reaction below to form ammonia: N2 (g) + 3 H2 (g) ⇌ 2 N...
A: the equilibrium concentration of NH3 can be calculated as
Q: I. Directions: Draw the lewis structures of the neutral atom and the ion of each element. Determine ...
A: Lewis’s structure: Lewis’s bonding theory is based on the octet rule. The Lewis structure is a simpl...
Q: The standard cell potential for the reaction at 298.15 K: Fe (s) + Zn2* (aq) = Fe2* (aq) + Zn (s) is...
A:
Q: The cell notation for the Galvanic cell is The Standard Reduction Potential are as follows: E°ca2*/c...
A: The cell given is Cd(s) | Cd2+ || I2 (s) | I-.
Q: Either sulfuric acid or bismuth(III) nitrate pentahydrate can catalyze this reaction. What is an adv...
A: Either sulfuric acid or bismuth(III) nitrate pentahydrate can catalyze this reaction.we have to desc...
Q: 2 CO(g) + O2(g) +2 CO2(3) K = 6.1 x 105 If [CO] = 0.00100M and [O2] = 0.00127 M, determine [CO2], in...
A: The given balanced chemical reaction is: 2 CO(g) + O2(g) <--------------> 2 CO2(g) The Kc expr...
Q: 15) Which one of the compounds corresponds to this IR spectrum. NH2 1000 2000 3000 4D00
A:
Q: product, determine the time for A to reach 1.438 M. Use the given: initial concentration of 2.553 M ...
A: For 1st order kinetics, kt = ln(Ao/A) Where, k = rate constant t = time Ao = initial concentrati...
Q: The AG°rxn for the reaction: N2(g) + O2(g) 2 NO(g) is +173 kJ/mol at 25 °C. What is the equilibrium ...
A:
Q: Create a model of the atoms of a substance moving through the solid, liquid, and gas states. Be sure...
A:
Q: The cell notation for the Galvanic cell is Cd(syCd2+/l/½(syI" The Standard Reduction Potential are a...
A: Given cell notation is : Cd (s) | Cd2+ (aq) || I2 (s) | I- (aq) E0Cd2+|Cd = -0.403 V (anode) E0I2|...
Q: explain the results of this experiment Table 1 Absorption Spectrum of Cobalt Chloride Using a Sp...
A: Given Wavelength Absorbance 400 410 420 430 440 450 460 370 480 490 500 510 520 ...
Q: 1. The positivity rate in a certain group is 3.5%. Determine the probability that a batch will test ...
A:
Q: A 12.g of methyl tert-butyl ether compound (contains C, H and O) was burned in an apparatus and 30....
A:
Q: The substance benzoic acid (C6H5COOH) is a weak acid (Ka = 6.3×10-5).What is the pH of a 0.258 M aqu...
A: 1) Given: Concentration of C6H5COOK = 0.258 M The hydrolysis reaction of C6H5COO- coming from C6H5CO...
Q: IV. Laboratory Report: (Parts 1,4,5 are typed.) 1. Title page including the average calculated value...
A: Given: Titration KHP (g) Vfinal (mL) Vinitial (mL) Vtotal NaOH(mL) 1 0.7430 33 0.0 33.00 2 0....
Q: Gaseous methane (CH,) will react with gaseous oxygen (0,) to produce gaseous carbon dioxide (CO,) an...
A: Given : Mass of CH4 = 0.96 g Mass of O2 = 7.53 g To calculate :- Mass of H2O (in grams)
Q: The following data were acquired in an iodination experiment involving acetone. Reaction times are f...
A:
Q: Consider the reaction: N2O4(g) 2 NO2(g). What is ΔS° at 25 °C? ΔHf°, kJ mol-1 ΔGf°, kJ mol...
A:
Q: What is polarity in chemistry
A: It is a physical property of compound which relates other physical properties such as melting point,...
Q: Your soap contains the glycerol which is the by-product of the hydrolysis reaction. You did not acc...
A:
Q: If the available number of valence electrons for a molecule or polyatomic ion does not complete all ...
A: If the available number of valence electrons for a molecule or polyatomic ion does not complete all ...
Q: 1. Particles of the reactants (1) must not only have enough energy upon collision for a reaction to ...
A: The collision theory describes that a reaction will take place when the reactant of the reaction cro...
Q: When is the fugacity equal to the partial pressure of a gas? O Fugacity is equal to partial pressure...
A: Given : We have to explain whether fugacity is equal to partial pressure of gas.
Q: when 15.00 mL of 2.00 x 10^-6 M lithium sulfate is mixed with 17.00 mL of 1.00 x 10^-6M sodium phosp...
A: Given: Concentration of lithium sulfate i.e. Li2SO4 = 2.00 × 10-6 M Volume of Li2SO4 solution = 15.0...
Q: Was there any physical change in the characteristics of trichloroethane when it was added to water?
A: Trichloroethane is micible in water. When it is added to water there is no any physical change.
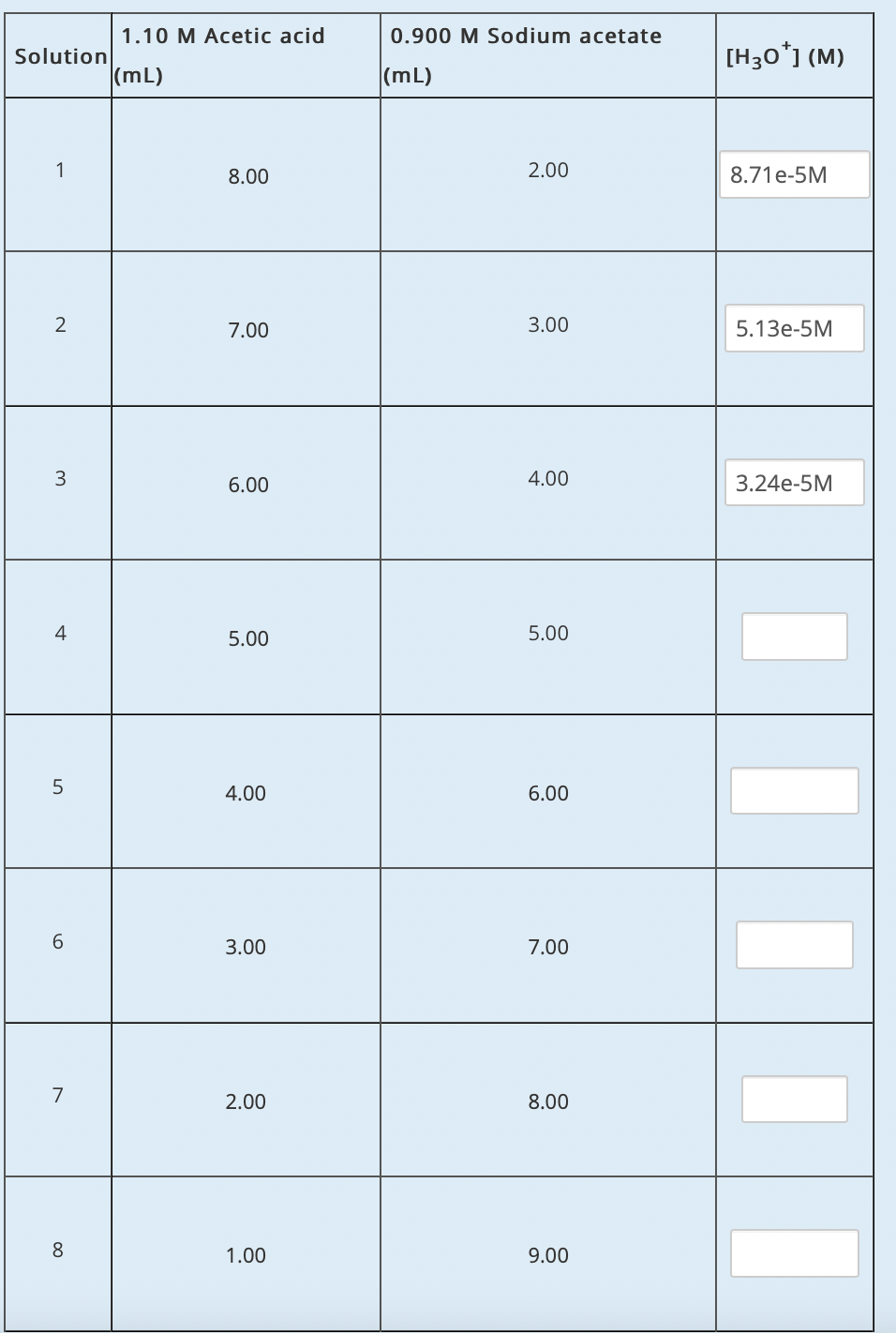
Q: Question 2 The molar conductivity at infinite dilution of hydrobromic acid, sodium acetate, & sodium...
A:
Q: A chemist analyzes a white solid compound and finds that it does not dissolve in water. When the co...
A: The polar or ionic compounds dissolves in polar water solvent. The covalent compounds are non polar ...
Q: How many grams of calcium phosphate contains 2.15 moles?
A: Given :- number of moles of calcium phosphate = 2.15 mol To calculate :- mass of calcium phosphate...
Q: he highest solubility in u
A:
Q: Nitric oxide, NO, is believed to react with chlorine according to the following mechanism: NO + Cl2...
A: Which of the following is/are and intermediate in the reaction?
Q: Materials and detailed methods: paragraph or two explain how the experiment was done Raw Data: W...
A: Diels -Alder reaction takes place in reactions between conjugated diene and an alkene. In this react...
Q: What is the product of the nucleophilic substitution reaction shown below? CH;SH Br HS H H SH II Mul...
A:
Q: -1490 wing molecule, iustrale the molecuia I (M*) first and then cations (M-N) and
A:
Q: When CH3COOH(ℓ) is dissolved in water, what covalent bonds break?
A:
Q: The acid H2CO3 decomposes to some extent in water to produce CO2 gas. What should you expect to obs...
A: The answer is as follows:
Q: At 100oC and 2 bar pressure, the degree of dissociation of phosgene is 6.30 x 10-5. Calculate the e...
A:
Q: An excessive amount of LiBr solid is added to water to form a saturated solution at 1 atm and 25 °C....
A: We would use the factors which affect the solubility of solid in water to check which process will i...
Q: Which of the following statement/s is/are TRUE? I. When electricity is passed through a solution con...
A: We are given four statement about the reaction occurring in electrochemical cell and we have to sele...
Q: 2s0,(g)+0,(3) 2s0,(g) ppose a mixture of SO,, O, and SO, has come to equilibrium in a closed reactio...
A: Given : We have to fill the table.
Q: How many grams of gas are present in each of the following cases? b. 8.75 L of C₂H₂ at 378.3 kPa and...
A: We have to predict the mass of gases.
Q: Which of the following statement/s is/are TRUE? The Arrhenius Theory cannot be applied to account ...
A: 1) statement 1 is correct beacuse Arrhenius Theory explain the variation of molar conductivity of we...
Q: Why are some metabolic reactions coupled to the hydrolysis of ATP? To bypass metabolic regulation To...
A: During the hydrolysis of ATP, the free energy is released.
Q: 3 mol % Bi(NO3)z*5H2O H,O, rt 3 mol % Bi(NO3)•5H,O H CH;CH,OH, rt
A: Given : We have to make the product for the given reactions.
Step by step
Solved in 2 steps

- If 500mL of 0.10M Ca2+ is mixed with 500mL of 0.10M SO42-, what mass of calcium sulfate will precipitate? Ksp for CaSO4 is 2.40 • 10-5. express answer to 3 significant figures. And include units.If a student combines specific amount CaCl2. 9H2O and Na2CO3 and obtained a solid precipitate. The student dissolve 1.37 gram of CaCl2. 9H2O in 25.0 mL of water and 0.984 g of Na2CO3 in 25.0 mL of water. The student is then combining the two solutions and observed the formation of a solid precipitate. The student filter the solution and recovered the precipitate. The following data were obtained for the experiment. Initial Mass of CaCl2. 9H2O 1.37 g Initial Mass of Na2CO3 0.984 g % yield of this experiment 87.4% 1. What is the balanced equation for this experiment?If a student combines specific amount CaCl2. 9H2O and Na2CO3 and obtained a solid precipitate. The student dissolve 1.37 gram of CaCl2. 9H2O in 25.0 mL of water and 0.984 g of Na2CO3 in 25.0 mL of water. The student is then combining the two solutions and observed the formation of a solid precipitate. The student filter the solution and recovered the precipitate. The following data were obtained for the experiment. Initial Mass of CaCl2. 9H2O 1.37 g Initial Mass of Na2CO3 0.984 g % yield of this experiment 87.4% Question 1. Determine the amount of the solid in (grams) that the student recovered in this experiment. Question 2. How many grams from the excess reactant remains unreacted in this experiment?
- Solid ammonium chromate is slowly added to 75.0 mL of a calcium acetate solution until the concentration of chromate ion is 0.0154 M. The maximum amount of calcium ion remaining in solution is M. Please answer asap Thanks!A 50.00 mL volume of 0.0600M K2CrO4 is mixed with 50.00 mL 0.0800 M AgNO3. Calculate the concentration of Ag+, CrO42-, K+, and NO3- at equilibrium. The solubility product of Ag2CrO4(s) is 1.20 x 10-12. Please show workOne mixes aqueous NaCl with aqueous AgNO3. NaCl. AgNO3and NaNO3all have appreciable solubility in water. AgCl has a solubility product constant Ksp of 1.6 x 10-10. If one mixes the NaCl solution with the AgNO3solution, one would suspect: a) no precipitate will form b) AgCl precipitate will form c) NaNO3 precipitate will form d) both AgCl and NaNO3 precipitates will form
- Find calculated k value per trial: Average k value: The initial volume of syringe was 15ml and I converted everything to L because that is what my manual said to do. I am confused with my excel spreadsheet as well. Am I doing my calculations correctly?Complete the following: 1. Random error, also called _________arises from the effects of uncontrolled variables in the measurement. 2. The ____________,measures how closely the data are clustered about mean. 3. The chemical equilibrium in which all the reactants and products are in the same phase are called____________. 4. Reaction that go to completion and never proceed in the reverse direction are said to be_____________ 5. The equation for buffer is the__________equation. 6. A ___________solution is one that resists changes in pH when small quantities of an acid or an alkali added to it. 7. __________is the pH of the pure, neutral and polyprotic1. The Ksp of Ca3(PO4)2 is 1.3 × 10−26. Estimate the solubility of this salt in units of g. L−12. If a sample of solid Ca3(PO4)2 is stirred into exactly one litre of a 0.550M solution of Na3PO4, how will the solubility of the salt compare with the answer that you have obtained in question 2.1? Explain you answer in a short sentence. Please only answer 2nd Question
- Calcium carbonate (e.g. from limestone) is relatively insoluble in water, and the solubilitydecreases with rising temperature. This is why CaCO3 precipitates out as ‘scale’ in hot water more readily than cold water. At a temperature of 20oC, calcium carbonate pKs= 8.8; at 35oC, pKs= 9.25. If you had water that was at equilibrium with excess calcium carbonate at 20oC and then raised the temperature to 35oC, what is the mass of CaCO3 that will precipitate out per litre of water? Hint: the difference in solubility will drive the reaction back to solid. CaCO3(s)↔Ca2++CO2−__K2S(aq) + __H3PO4(aq) ® __K3PO4(aq) + __H2S(g) In a reaction mixture of 14.2 mL of 0.42 M K2S and 6.0 mL of 0.85 M H3PO4: 1. The theoretical yield of H2S from this reaction mixture, in mL at STP is _______ mLSTP 2. If 120 mL at STP of H2S was isolated, the percent reaction yield is ______Solid cobalt (II) acetate is slowly added to 125 mL of 0.0945 M ammonium chromate solution. What is the concentration of cobalt required to just initiate precipitation? The Ksp of CoCrO4 is 7.1 * 10 -4. Report answer in scientific notation to two sig figs.



