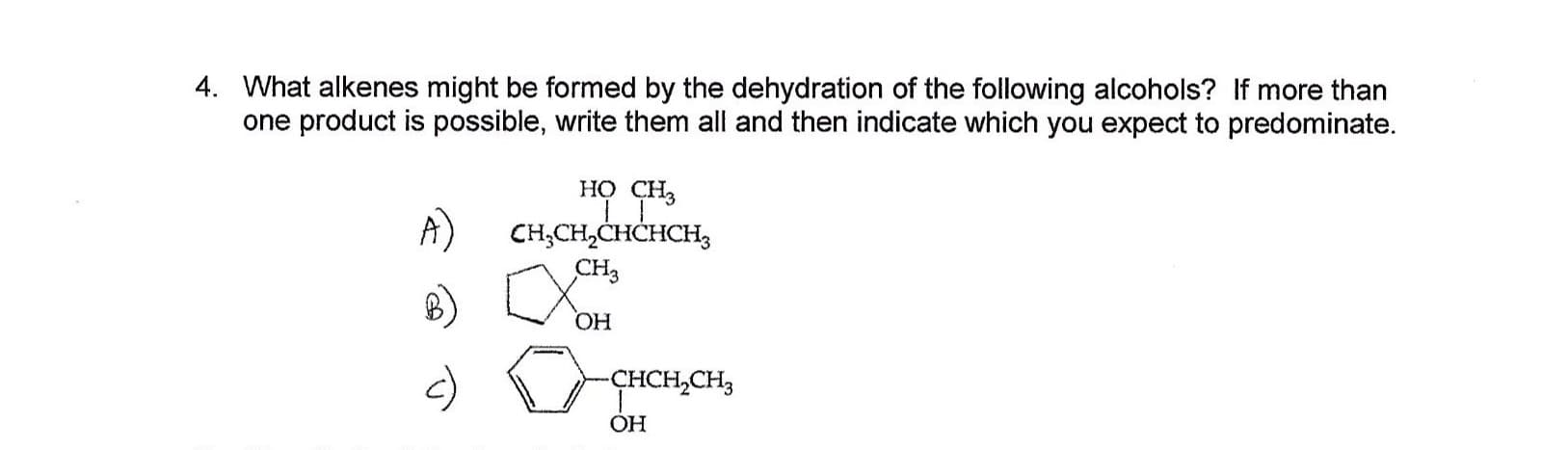
4. What alkenes might be formed by the dehydration of the following alcohols? If more than one product is possible, write them all and then indicate which you expect to predominate. но сн, A) CH,CH2CHCHCH3 сHз ОН -ҫнCH,CH, OH
Q: Draw a structural formula for the alkene with the molecular formula C5H10 that reacts with Br2 to…
A: The Bromination of alkene occurs through an intermediate bromonium ion. The compound 1,…
Q: When 2-bromo-2,3-dimethylbutane reacts with a strong base, two alkenes (2,3-dimethyl-1-butene and…
A: An E2 reaction is a concerted, one-step reaction in which the proton and the halide ion are removed…
Q: НО SH CH2OH NC
A:
Q: What are the major and minor products formed when cyclohexene reacts with Br2 in the solvent CH2Cl2?
A:
Q: An unknown alcohol with a molecular formula of C7H14O was oxidized to an aldehyde with HOCl. When an…
A: Molecular formula of the given alcohol is C7H14O. According to the given question:
Q: Complete the following reaction by drawing a structural formula for the product. CH2CH, (1) NABH4…
A: Finding the product of the above reaction.
Q: What alkenes might be formed by dehydration of the following alcohols? CH3 CH,CH,CH,ČCH,
A: In dehydration of alcohol water is removed from compound and alkene is formed.
Q: Write the complete cquation for the dehydration reaction of the following alcohols. If more than one…
A:
Q: a) ethane with 1₂ b) cyclohexane with Cl₂ c) methylcyclohexane with Cl₂ d) methylcyclohexane with…
A:
Q: a Draw structural formulas for all possible carbocations formed by the reaction of each alkene with…
A:
Q: An unknown alcohol with a molecular formula of C7H14O was oxidized to an aldehyde with HOCl. When an…
A:
Q: c=CH H2O, H2SO4 H9SO4 `CH3 Alkynes do not react directly with aqueous acid as do alkenes, but will…
A:
Q: Complete the equation for the following reaction by drawing a structural formula for the missing…
A: When a carboxylic acid, 2-methylbutanoic acid is treated with ammonia then 2-methylbutanamide is…
Q: Propose a structural formula for the product(s) when each of the following alkenes is treated with…
A: Answer -
Q: When 2-bromo-2,3-dimethylbutane reacts with a strong base, two alkenes (2,3-dimethyl-1-butene and…
A: An E2 reaction is a concerted, one-step reaction in which the proton and the halide ion are removed…
Q: Which alkene would the following alcohols form upon dehydration reaction using POCl3 in pyridine?…
A:
Q: D. Give the products that would be formed when each of the following alcohols is subjected to…
A: The reaction that converts the alcohol functional group into an alkene is termed as the dehydration…
Q: 5. Which of the following would not yield the alcohol shown below? он а. 1. „MgBr 2. H;O* b. 1.…
A: An organo Lithium Reagent is R-Li and a Grignard Regent is R-MgX, R is an alkyl group and X is a…
Q: Draw structural formulas for the alkenes formed on acid-catalyzed dehydration of each alcohol. Where…
A: In acid catalyzed dehydration of alcohols, alkenes are formed.
Q: 1. Reaction of 2,3-dimethyl-1-butene with HBr leads to an alkyl bromide, C6H13Br. On treatment of…
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: Draw structural formulas for the alkenes formed on acid-catalyzed dehydration of each alcohol. Where…
A: Alcohols undergo dehydration reaction through acid-catalyzed process to produce alkenes. Alcohols…
Q: Draw structural formulas for the alcohol formed by hydroboration-oxidation of each alkene.
A: In a hydroboration-oxidation of alkene, Alkenes reacts with the BH3 followed by H2O2 and NaOH.…
Q: Draw a structural formula for the alkene with the molecular formula C5H10 that reacts with Br2 to…
A: The halogenation reaction is the reaction which gives product with one or more halogens attached.…
Q: Prednisolone is a corticosteroid used to treat certain types of allergies, inflammatory conditions,…
A:
Q: Propose a structural formula for the product(s) when each of the following alkenes is treated with…
A: When alkene react with water in presence of Sulphuric acid, it gives alcohol. This process follows…
Q: Draw a structural formula for the alkene with the molecular formula C5H10 that reacts with Br2 to…
A: In the addition reaction of bromine, the alkene react with Bromine solution in presence of…
Q: The ester with the formula C8H1602 gives an alcohol and an acid when hydrolyzed. When the alcohol is…
A:
Q: Give the structure of an alkene (or alkenes) with the same number of carbon atoms for the synthesis…
A: Since you have posted a question with multiple sub-parts,we will solve first three sub-parts for…
Q: 9. There are several possible forms of a trisubstituted cyclohexane with the formula C10H200. I have…
A: To identify the most and least commonly naturally occurring compound.
Q: 1. Draw the structural formulae of the products formed when 1-butene reacts with each of the…
A: When treated with HX alkenes form alkyl halides. Hydrogen halide reactivity order : HI > HBr >…
Q: 2. Complete the following reactions for the preparation of alcohols. Draw the structure of the…
A:
Q: Draw the structural formula of the products formed when each alkene is treated with one equivalent…
A: INTRODUCTION: given structure: Pent-2-ene The full form of NBS is…
Q: (a) Which carbonyl compounds would CH3 CH3CH,CH2CH,CCH,OH (b) HyÇ OH (e) you reduce to produce the…
A: The compounds given are,
Q: What is the general format of a reagent for the reaction of an alkene or alkyne to count as…
A:
Q: Draw a structural formula of an alkene or alkenes (if more than one) that undergo acid-catalyzed…
A: The given product is 2-methyl-2-butanol.
Q: Draw the structures of the chief product formed when the following alcohols are dehydrated to…
A: the structures of the chief product formed when the following alcohols are dehydrated to alkenes are…
Q: Draw the structure of the product of this reaction. H CH2CH3 Br H-.. КОН E2 elimination product H--…
A:
Q: 3. The following alkenes can be prepared by dehydration of an appropriate alcohol. Show the…
A: Hello ! Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the…
Q: Which are secondary alcohols? H. CH; CH3 H,C H,C-C-OH H `CHCH, OH CH3 II III IV O A. I, II B. II, II…
A:
Q: Which structures have the correct IUPAC names? CH3 CH3 Br Br I. 2-bromotoluene II. para methyl, meta…
A: The compounds given are,
Q: 9) For the reaction between isopropyl 1-propyl (or 'n-propyl') ether and HBr, what are the expected…
A: option A
Q: 5. Which would have the higher boiling point. Explain your answer: Propane or 2-chloropropane (1) 6.…
A: 5. Between Propane and 2- chloropropane 2-chloropropane has higher boiling point because of…
Q: Draw structural formulas for the alkenes formed by acid-catalyzed dehydration of each alcohol. Where…
A:
Q: 5 a Copy and complete the two equations below, which can be used to show the alkylation and…
A: Detail description is given below
Q: What alkene is needed to synthesize each 1,2-diol using (1) "OH in H,0? CH,CH,CH, но OH a. CH,CH,CH,…
A:
Q: OH H,SO4, 180°C
A: Applying concept of E1 elimination reaction.
Q: Alkenes can be converted to alcohols by reaction with mercuric acetate to form a…
A: Alkenes will be converted to alcohols on reacting with mercuric acetate.
Q: Alkenes can be converted to alcohols by reaction with mercuric acetate to form a…
A: In the oxymercuration reaction, the addition of Hg(CH3COO)2 and OH occur via the anti-addition…
Q: TRUE OR FALSE I. The reaction of BH3 with 2-methylbut-2-ene results in the attachment of a hydroxyl…
A: TRUE OR FALSE
Q: 1) 3- Methyl-2-butanol will react with H2SO4 to give two isomeric alkenes. Write suitable reaction…
A: Given Reactant = 3 methyl 2 butanol and H2SO4 Major Product = IUPAC name of each product = ?
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 4 images

- Propose a structural formula for the product(s) when each of the following alkenes is treated with H2O/H2SO4. Why are two products formed in part (b), but only one in parts (a) and (c)? (a) 1-Hexene gives one alcohol with a molecular for- mula of C6H14O. (b) 2-Hexene gives two alcohols, each with a molecu- lar formula of C6H14O. (c) 3-Hexene gives one alcohol with a molecular for- mula of C6H14O.How many alkene products, including E, Z isomers, might be obtained by dehydration of 3-methyl-3-hexanol with aqueous sulfric acid?Draw structural formulas for the alkenes formed on acid-catalyzed dehydration of each alcohol. Where isomeric alkenes are possible, predict which alkene is the major product. Q. 2-Methylcyclopentanol (racemic)
- 1. Predict the elimination products of the following reactions. When two alkenes are possible, predict which one will be the major product. Explain your answers, showing the degree of substitution of each double bond in the products. 2. Which of these reactions are likely to produce both elimination and substitution products? (a) 2-bromopentane +NaOCH3 (b) 3-bromo-3-methylpentane +NaOMe(Me= methyl, CH3) (c) 2-bromo-3-ethylpentane +NaOH (d) cis-1-bromo-2-methylcyclohexane +NaOEt (Et= ethyl, CH2CH3)In each case below select the synthetic procedure/s that could be used to carry out the transformation, giving the alcohol shown as the single major product.The procedures are:Hydroboration/oxidation: alkene + BH3; then H2O2, -OH.Oxymercuration: alkene + Hg(OAc)2, H2O; then NaBH4When 2-pentene is treated with Cl2 in methanol, three products are formed. Account for the formation of each product (you need not explain their relative percentages).
- 1. Ethylene glycol, a major component of antifreeze, becomes especially poisonous when it is oxidized by the liver. Draw two oxidation products that could be derived from ethylene glycol. 2.When 2-methyl-1-butanol is dehydrated in an acid medi- um to an alkene, it yelds mainly 2-methyl-2-butene rather than 2-methyl-1-butene. This indicates that the dehydra- tion to an alkene is at least a two-step reaction. Suggest a mechanism to explain the reaction.Alkenes can be hydrated to form alcohols by (1) hydroboration followed by oxidation with alkaline hydrogen peroxide and (2) acid-catalyzed hydration. Compare the product formed from each alkene by sequence (1) with those formed from (2). Q.)trans-2-ButeneWrite the structure of the compound E,E-2,4-Hexadien-1-ol and label each non-equivalent carbon with a letter, A,B,C..
- Draw a structural formula for the alkene with the molecular formula C5H10 that reacts with Br2 to give each product.A chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2 CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under the same conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C. Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structures of A, B, and C; give equations for their formation; and explain the stereospecificity of these reactions.draw the two possible carbocations that can form when this alkene reacts with a strong acid (such as HBr or H3O+). of the two structures you drew, circle the more stable carbocation

