Q: 1. What type of product was formed in A.1.a based on the color of the litmus aper?
A: The compounds which turn blue litmus red are called acids. They taste sour and have a pH lower than…
Q: For the reaction below, Kc = 0.60 at 550 K. The reaction starts with a 0.050 M concentration of…
A: Given : Kc = 0.60 Initial Concentration of A2B2 = 0.050 M Temperature = 550 K
Q: Indicate at which position(s) the following electrophilic aromatic substitutions will take place and…
A: two reaction are given , we have to tell where electrophile will attack with showing resonance
Q: After analysis for iron content, a water sample was found to have a concentration of 4.62ppm iron.…
A:
Q: 3. Sucrose has the chemical formula of C12H22011. How many moles of each type of atom are in 1 mol…
A: In one mole of any substance there are avogadro's number of entities. Given number of moles of…
Q: 1. Give the IUPAC name for each compound. a. NC. b. you C. Br
A:
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: We have been given reactants and products.We have been asked to draw curved electron pushing arrows…
Q: Linear density is a measure of how much a crystallographic vector is occupied by atoms, and is…
A: a. The lengths of the [100] and [110] vectors for an FCC (face-centered cubic) crystal are equal to…
Q: Which one is more concentrated? 1M or 1m of the solution? Give a reason. Cite at least three…
A: Check which solution is more concentrated and mention the reasons for it.
Q: 1. The original concentration of a solution is 200 mg/dL. The solution was diluted 1:5. Then one…
A: Give, The initial concentration of a solution = 200 mg/dL. Here, 1:5 indicate, one part of the…
Q: Write the balanced COMPLETE ionic equation for the reaction when CaCl₂ and Na₃PO₄ are mixed in…
A: Since, Balanced reaction means that both side number of atom present in equal number. Thus, For…
Q: A 0.238 g0.238 g sample of a metal, M, reacts completely with sulfuric acid according to…
A:
Q: Write a balanced net ionic equation to show why the solubility of Co(OH)2(s) increases in the…
A: Answer: For a sparingly soluble compound it is considered that all the dissolved quantity of the…
Q: ●●● PRACTICE PROBLEM 11.8 Show how you would prepare the following compounds from the appropriate…
A: We have find out the answer.
Q: Cite at least two applications of concentrations that you do at home and explain why does it…
A: Concentration refers to the amount of a substance that is present in a given volume or mass of a…
Q: НО. CI
A: Answer :
Q: Give the structure that corresponds to the following molecular formula and ¹H NMR spectrum::…
A: Given that, the molecular formula is C7H16O4. We have to determine the structure of the compound.…
Q: Linear density is a measure of how much a crystallographic vector is occupied by atoms, and is…
A: a. The lengths of the [100] and [110] vectors for an FCC (face-centered cubic) crystal are equal to…
Q: 1. Write the reaction quotient and the equilibrium expression for the following reactions: a.…
A: The reaction quotient (Q) and the equilibrium expression (K) are both used to describe the state of…
Q: 8.17) What is the acid form of NH3? Như NH3 + H - NHu = Proton Acid is NH3-base Sa a proton (Ht)…
A: Answer :
Q: In general, what does the %error tell you about the expected results in the experiments for Silver…
A: The %error (percent error) is a measure of how accurate an experimental result is compared to the…
Q: a chemist determines that 1.26gof iron reacts with 0.54gof oxygen to form rust. whatis the percent…
A:
Q: Determine the pH change when 0.084 mol HCIO4 is added to 1.00 L of a buffer solution that is 0.342 M…
A: Answer :
Q: o= OH Please help me name this!!!
A: IUPAC name of an organic compound contains prefix, word root and suffix. Prefix is for the…
Q: Calculate how many kilograms of SiO2 are required to produce 3.67 kg of SIC?
A: The given information is as follows: The mass of SiO2 required = ? The mass of SiC…
Q: Br3:05Up 40 °C + and se
A: Answering only the first question as per guidelines for further assistance repost them seperately.
Q: 2. Potassium chlorate decomposes to produce potassium chloride and oxygen. If 1.50g of potassium…
A:
Q: C²H+H $ двина I O С CH3
A: Answer :
Q: Write Lewis Formula including resonance forms for SO2 and SO3
A:
Q: name both c and d H₂C C. H₂C d. CH₂ CH₂1 CH 3 CH3 CH 3 CH₂ CH₂ \ CH3
A: Number of C-atoms. Name 1 meth 2 eth 3 Prop 4 But 5 Pent 6 Hex 7 Hept If all C-C bond…
Q: HNMR Spectroscopy.. Please solve and explain it
A: Now, we have to calculate the double bond equivalence (DBE) by using the formula for molecular…
Q: What is the structure of SeF5NF2?
A:
Q: A serum is diluted 1:20 then 2 parts of the resulting dilution is added to 40 parts saline. Then 3…
A: To solve this problem, we need to calculate the total dilution factor of all the dilutions. The…
Q: How do I find initial concentration of iodine in reaction and initial concentration of thiosulfate…
A:
Q: 5. 6 CH3 10 H СЕ Et Me Be ? C²H&H Ⓒ
A: With grignard reagent ring opening of epoxide takes place in anti manner followed by hydrolysis…
Q: 23. A researcher wanted to use myoglobin as a substitute for hemoglobin to carry oxygen in a human…
A: We from the overall observation of the graph we can say the myoglobin curve is hyperbolic and the…
Q: A solution with 41.2 mol HCI is mixed with a solution with 48.8 mol NaOH. The volume is 712 L. What…
A: Answer: Given electrolytes in the problems are strong electrolytes, therefore when added in water…
Q: A robot spacecraft returned samples from the planetesimal 98765 ALEKS, located in the outer Solar…
A: Answer: When two atom have same atomic number but different mass number then they are called as…
Q: Mass of Al sample: 0.045g Vol. of gas collected: 45.38ml Barometric Pressure : 755 mmHg Height of…
A: Given , mass of AI sample = 0.045 g Height of water column = 12.45 cm H2O…
Q: Gaseous methane CH4 will react with gaseous oxygen O2 to produce gaseous carbon dioxide CO2…
A:
Q: QUESTION 2 Calculate the AG in kJ of the following reaction under standard conditions: 2AI + Fe2O3…
A:
Q: 2.) What is the pH of a 0.010M solution of pyridine, C₂H5N, a weak base? It's K is 1.5x10-⁹. SHOW…
A: The question is based on the concept of chemical equilibrium. we need to calculate pH of the given…
Q: Calculate the molar solubility of BaSO4 in a solution in which H3O+ is (Ksp (BaSO4) = 1.1 x 10-¹0,…
A: To calculate the molar solubility of given salt in different Molarity of given acid. Solving the…
Q: What is the IUPAC name for the compound shown?
A:
Q: 2C1O₂(aq) + 2OH(aq) [CIO₂] (M) 0.0500 0.100 0.100 a. CIO3(aq) + CIO₂ (aq) + H₂O(1) [OH-] (M) 0.100…
A:
Q: proposed multiplication or division (5.0 cm²) (9.0 cm) = ? 2 60. mg 0.020 g = ? Is this possible? O…
A: given three multiplication and division , we have to tell whether it is possible to carry out…
Q: Question If we calculate the change in enthalpy of a reaction by using heat of formation data, the…
A:
Q: Consider the reaction: A → B. Use the below data to determine the first half-life for experiment 2.…
A: Answer: Power of the concentration term of a reactant in rate law is the order of reaction with…
Q: From the table of available reagents select the one(s) you would use to convert 1-phenylethanol to…
A: Alcohols converted into ether by treating with sodium amide to form the sodium alkoxide. Then sodium…
Q: Determine the number of atoms of O in 44.5 moles of Cr,(PO)₂
A:
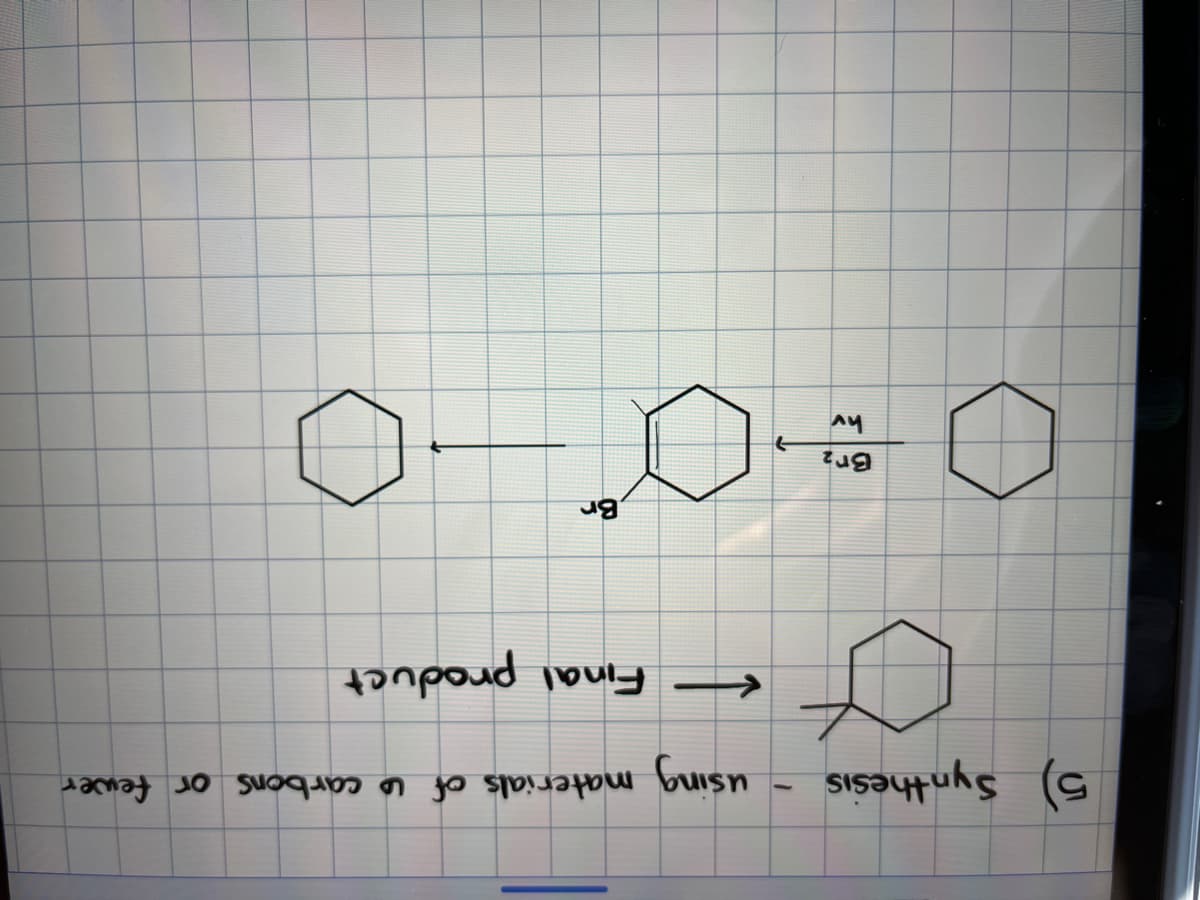
Step by step
Solved in 2 steps with 1 images

- Complete the reaction schemes below providing the products of the reaction schemes below.Find the product by writing down the mechanism of the reactions güven below.Sodium borohydride (NaBH4) can be used to reduce cyclohexanone to cyclohexanol. How could one us IR to determine if all the starting material has been consumed?
- Predict the product of the following reaction and classify the reaction Pb+FeSO4---> PbSO4+ ______What is the product of the following reaction? (please provide detailed steps on how you got to the answer) thank you :)Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- Rate = k [CH3CH2Br][CH3COO-] Which mechanism would best fit the data?

