Q: 5. A sample of gas has a volume of 215 cm3 at 23.5°C and 84.6 kPa. What volume will the gas occupy…
A: Given that, P1 =84.5 kPa =0.834 atm V1 =215 cm3 =0.215 L T1 23.5oC =296.5 K P2 =1 atm T2 =273 K
Q: When cooled from 248 °C, the diameter of a metal sphere becomes 72.0 mm and its surface area changes…
A:
Q: How much in peso is the cost of boiling 13.2 L of water from 22 °C using a heavy cook pot with mass…
A:
Q: A piece of 0.0332 kg mercury metal reaches its final temperature at 453.536oF with the addition of…
A: Heat capacity is the energy required to raise the temperature of given material by 10C. It is…
Q: How much heat is lost if 60 g of iron is cooled from 80 °C to 15 °C? The specific heat capacity of…
A: The heat lost is given by H = m c (T' - T) where, m is the mass, c is the specific heat and T' - T…
Q: A steel plate has an area of 2.09 m2 at 195 °C. Calculate its area when cooled to 10°C. [2.08 m2]
A: Given, Ti=195C , Tf= 10C Ai= 2.09m2 we need to find Af.
Q: How much heat in kcal must be added to 0.43 kg of water at room temperature (20°C) to raise its…
A:
Q: The classroom temperature is 20° C. The relative humidity is 60%. What is the total humidity (kg of…
A: The classroom temperature (T),
Q: What mass of water will change its temperature by 3 °C when 525 J of heat is added to it? The…
A: Given data:
Q: A Styrofoam box has a surface area of 0.84 m2 and a wall thickness of 2.0 cm. The temperature of the…
A:
Q: How many CAL must be added to heat 300.0 g of water from -37.0°C to 50.0 °C ? Don't do the…
A:
Q: A bar of 20.0 grams of aluminum of specific heat 0.215 cal/gramCo is heated from 15.0 Co to 20.0 Co.…
A: Given that, m=20.0 gc=0.215 cal·gram-1·°C-1Ti=15.0 °CTf=20.0°C
Q: What quantity of heat is required to raise the temperature of 450 grams of water from 15°C to 85°C?…
A:
Q: As the temperature increases, which of the following statements is correct? * (1 Point) b. Specific…
A: Density is m/V (mass over volume); if volume increase, density decrease. Thus an increase of…
Q: What mass of water can be heated from 25.0° C to 50.0° C by the addition of 2825 J? * 27 gm O 22 gm…
A:
Q: A brass plug has diameter 10.0 cm @ 25°C. To what temperature must the plug be chilled with dry ice…
A:
Q: An incandescent light bulb with a surface area of 0.0131 m2 and an emissivity of 0.95 has a surface…
A:
Q: A 681-g empty iron kettle is put on a stove. How much heat, in joules, must it absorb to raise its…
A:
Q: Your 27 °C bedroom uses a spherical bulb operating at a temperature of 2794 °C. Assuming that the…
A:
Q: If it takes 41.72 joules to heat a piece of metal weighing 18.69 g from 10.0 °C to 27.0 °C what is…
A:
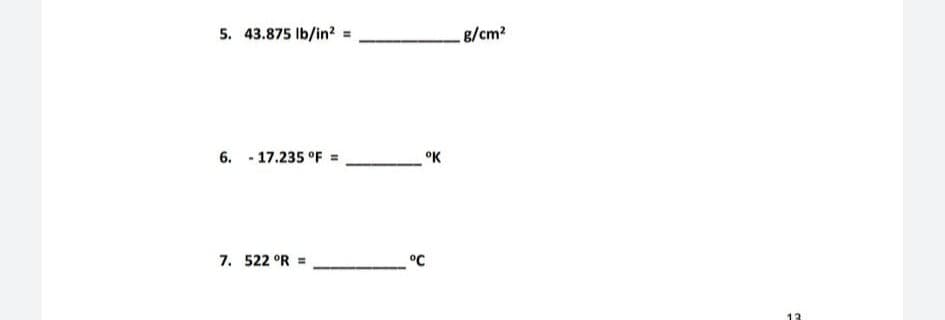
Q: Express the temperature (73.2oF) in Celsius degrees. Please include 1 decimal place in your answer.
A: The temperature in degree Celsius and Fahrenheit are related as C=59F-32
Q: A 242.0 g china serving bowl is warmed 65.9°C when it absorbs 3.3 kcal of heat from a serving of hot…
A: we know heat flowing through a material Q = Cm∆T where C = specific heat m = mass of substance ∆T=…
Q: made of copper (c=0.0923 cal/g-C°) of mass 300 g contains 450 grams
A:
Q: What will be the temperature change of a 0.5kg iron block when 1125J of heat is added to it? 5°C O…
A:
Q: 11. A steel rod with 12.97 cm area and a steel ring at 11.00 cm both at 25 C must be heated or…
A:
Q: 2. Compute the amount of heat per second through an iron plate 2 cm thick and area of 0.5000m2. If…
A:
Q: Specific heat capacity of ice is 0.5 cal/g °C. What will be this value in J/kg °C а. 8761298 O b.…
A: It is known that 1 cal/g °C = 4186 J/kg °C .
Q: 17) On a cold day, a piece of metal feels much colder to the touch than a piece of wood. This is due…
A: Thermal conductivity of metal is more than thermal conductivity of wood . therefore we feel much…
Q: How much heat in kcal must be added to 0.58 kg of water at room temperature (20°C) to raise its…
A:
Q: 2.6 of water at 28 °C is added to 5.0 of water at 69 ° C. What is the temperature of the mixture in…
A: Here the unit of mass is not provided. However since the unit of specific heat is provided in si…
Q: "A 0.25 kg length of aluminum wire is warmed 10.0°C by an electric current. How much heat was…
A:
Q: Temperature Measurements Scale 1 (°C) Scale 2 (°C)Average (°C) Average (°F) Average (K)% Difference…
A: The Celsius scale to Fahrenheit scale conversion is as follows The Celsius scale to Kelvin scale…
Q: A piece of an unknown solid substance weighs 437 g and requires 8460 J to increase its temperature…
A:
Q: A circular 1.5 kg aluminum plate with a radius of .153 m and a thickness of 0.75 cm is in contact…
A: mass =1.5 kg radius = 0.153 m thickness = 0.75 cm = 0.0075 m change in temperature = 63-19 = 44 C…
Q: A 95.0 g sample of metal is warmed 26.0 degrees * C when 65.0 calories is added. What is the…
A: Given: The mass of the metal is 95 g. The increase in the temperature is 26°C. The heat added to the…
Q: A 652-g empty iron kettle is put on a stove. How much heat, in joules, must it absorb to raise its…
A:
Q: One liter of a certain gas is heated from 25 degrees C to 1000C. What is the final volume?β= 950…
A:
Q: When 200 mL of water in an electric kettle is heated from 20°C to 35°C to make a cup of tea, how…
A:
Q: The vapor pressure of water at 5°C is 0.8726 kPa. Calculate the vapor pressure in atm.Round answer…
A: Converting the pressure in kPa to Pa, 0.8726 kPa=0.8726 kPa×1000 Pa1 kPa=872.6 Pa
Q: How much heat is released when 50g of water at 76°C cools to 15°C? The specific heat of the water is…
A: Given(s): Mass of the water, m=50 g=0.050 kg Change in temperature, ΔT=76-15=61o C Specific heat of…
Q: specific
A: A substance that has more specific heat absorbs a large amount of heat without rising in…
Q: 8. A 1.50 × 10² g piece of brass (specific heat capacity 3.80 x 10² J/(kg-°C)) is submerged in 400.0…
A: The net heat loss of the system is zero as there is no heat transfer to the surrounding. Here, cw…
Q: 273 cm³ of an ideal gas has an initial temperature of 0°C. The gas is heated at a constant pressure…
A: According to Ideal gas law volume is directly proportional to temperature at constant pressure.i.e.,…
Q: Calculate the thermal conductivity of a wood used in 1.22m x 1.22m test panel, 2.54 cm thick if the…
A: Given : Power, P=dQ/dt = 36 W Thickness, x = 2.54 cm = 0.0254 m Area, A = 1.22 m × 1.22 m…
Q: Specific heat of an unknown substance is given as 0.86 cal/g°C.
A: The correct option is D. 3600
Q: How many grams of ice at a temperature of 0°C can be melted with 640 cal?
A:
Q: Specific heat of an unknown substance is given as 0.86 cal/g°C. What is the value of specific heat…
A: Given Data: Specific Heat of an unknown substance = 0.86 cal/g C To calculate : specific heat of…
Step by step
Solved in 2 steps with 1 images

- 1. A container used to store ice is made of wood 5.0 cm thick and has an effective area of 12,000 cm2 and thermal conductivity of 0.00027 cal/cm2 -sec-C/cm. How much ice inside the box would be melted each 24-hour day if the outside temperature is 25C and the temperature inside the box is 5C. Include FBD1.4L of vegetable oil is being heated. Vegetable oil contains an average density of 930kg/m3 and a specific heat of 1670J/kg degrees C. How much heat was transferred to the oil if it was heated from 21 degrees C to 90 degrees C? (1L=0.001m3)how much heat is lost when a 640 g piece of copper cools from 375oc ,to 26oc?(the specific heat of copper is 0.38452 j/goc)
- A piece of an unknown solid substance weighs 437 g and requires 8460 J to increase its temperature from 19 degrees C to 68 degrees C. What is the specific heat of the substance?When cooled from 248 °C, the diameter of a metal sphere becomes 72.0 mm and its surface area changes by 88.4 mm2. If α = 11.5 x 10-6 / °C, what is the final temperature? [13.3 °C ]A spherical steel ball bearing has a diameter of 2.540 cm at 25.00OC. What temperature change is required to increase its volume by 1.000%?
- One liter of a certain gas is heated from 25 degrees C to 1000C. What is the final volume?β= 950 x10-6/°C41. Determine the elongation of steel rod which is 100 meter in length if the temperatureincreases from 27oC to 57oC. Use linear coefficient of expansion of α = 11X10^-6/oC A. 3 mB. 23 mC. 2 mD. 33 mmWhat quantity of heat is required to raise the temperature of 450 grams of water from 15°C to 85°C? The specific heat capacity of water is 4.18 J/g/°C.
- 2. What mass of water will change its temperature by 3 celcius when 525 J of heat is added to it? The specific heat of water is 4180 J/kg Celsius1. A thermometer reads 30.9 °C. Calculate this temperature in Fahrenheit degrees. Fahrenheit temperature = 2. A thermometer reads 53.1° F. Calculate this temperature in Celsius degrees.Celsius temperature = 3. Which is higher 82.3 °F or 28.9 °C ?What is the specific heat of a 5.1 -kg substance if 135j of heat is needed to raise the temperature from 283.15 K to 303.15 K? A. 0.0018 B. 1.3 C. 0.76 D. 0.029

