Chapter7: Alkenes: Structure And Reactivity
Section7.SE: Something Extra
Problem 24VC: The following carbocation is an intermediate in the electrophilic addition reaction of HCl with two...
Related questions
Question
Can you help me answer 5.31
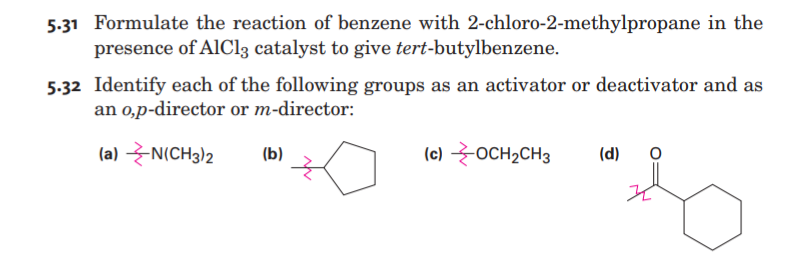
Transcribed Image Text:5-31 Formulate the reaction of benzene with 2-chloro-2-methylpropane in the
presence of AlCl3 catalyst to give tert-butylbenzene.
5-32 Identify each of the following groups as an activator or deactivator and as
an 0,p-director or m-director:
(a) N(CH3)2
(b)
(c) OCH2CH3
(d)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

