Q: KOH(aq) + HBrO (aq) KBrO (aq) + H2O (1) 2CH3(CH2) CH3(9) +1302(9) 8CO2(g) + 10H₂O(9) CH2CH2OH (1)…
A: Combination reaction:If 2 reactants combine to give the product, it is combination reaction.Single…
Q: Given the following information, construct a Born-Haber cycle to calculate the lattice energy of…
A: The Born-Haber cycle is a thermochemical cycle that represents the energies associated with each…
Q: Draw all significant resonance structures for each of the following compounds. (a) OH & (b) (c)
A:
Q: Polymers may be composed of thousands of monomers. Draw three repeat units of methyl methacrylate.…
A: Given that the reaction is:
Q: Which of the following four 13C NMR spectra belongs to phenol? 160 140 120 80 60 40 20 ppm 200 180…
A: 13C-NMR is used to obtain information about the carbon backbone of the compound chemical structure.…
Q: Be sure to account for all bond- breaking and bond-making steps. HH ΚΘ Br Select to Add Arrows…
A: Curved arrows show transfer of electrons from electron rich species (species containing loan pair or…
Q: barium chloride (aq) + sodium sulfate (aq) Balanced Formula Equation:
A: this is a type of double displacement reaction in which ions of both the salts are getting…
Q: 3. Provide a mechanism for the following transformation: H3C i HCI CH₂OH H3C Досна OCH3 HO
A: “Since you have posted a question with multiple questions, we will provide the solution only to the…
Q: What is the relationship between the parent structure on the left and the structure on the right? ..…
A: Compounds with the same molecular formula but different structures are known as isomers.Structural…
Q: Macmillan Learning For a particular isomer of CH₁g, the combustion reaction produces 5108.7 kJ of…
A: The isomer is of C8H18 The combustion reaction is as follows:C8H18(g)+25/2…
Q: Which nitrogen atom is most basic? CI CI О 20 a N CI N CI 8
A: Basicity refers to the chemical property of a substance that can accept protons (H+ ions) in a…
Q: a) PhCOOH b) c) 요 PhCOOH Ts DO (H3CO2P ✓✓ OCH3 |0
A: First two reactions are Baeyer-Viliger reactions and the second two reactions are Wittire…
Q: 12. A Zinc and Copper half cell were connected under standard conditions. Calculate the standard…
A:
Q: A 500 mL solution of 0.250 M NH, is required. What is the volume of 28wt \% of NH, (density = 0.899g…
A: The objective of the question is to determine the volume of of .It is given that,The volume of the…
Q: What is the predicted product of the following reaction? CI- CH3ONa NO2 I H3CO- -OCH3 CI OCH3 III…
A: Given ,Reaction : Objective : Determine the product of the reaction.
Q: The
A: To find the pH at which seawater, amorphous silica, and sepiolite are all in equilibrium, we need to…
Q: Draw the mechanism, including all the curved arrows, for each step of the following reaction:…
A: To draw the mechanism for the given transformations
Q: (B) Draw two chair conformations of the following substituted cyclohexane and identify the most…
A: In chair conformation of cyclohexane the bulkier group (ethyl) is present at equatorial position It…
Q: The following initial rate data are for the reaction of mercury(II) chloride with oxalate ion: 2…
A: General Chemistry question Explanation:Approach to solving the question: General approach Detailed…
Q: A solution NH3 that contains 78 mL of 0.043 M ammonia, NH 3, is titrated with 0.083 M HCI. The K₁ of…
A: Given,Molarity of NH3 = 0.043 Mvolume of NH3 = 78 mLMolarity of HCl = 0.083 MKb of NH3 = 1.8 x 10-5
Q: 14 What is the active electrophile in the EAS iodination of benzene with I₂ and HNO3 ? C a) III 6)…
A: Electrophiles are positively charged or neutral species.Electrophiles have empty…
Q: Add curved arrow(s) to draw step 6 of the mechanism. Modify the given drawing of the products as…
A:
Q: (E)-1,3,5-hexatriene B D Multiple Select Question Select all that apply Select all of the following…
A: It refers to the compounds having the same molecular formula, and bonding of atoms but differing in…
Q: Identify the type (sigma or pi) of bond and the total number of times it appears in the structure.…
A: A carbon atom forms four covalent bonds. Sigma covalent bonds are formed by the hybrid orbitals and…
Q: Determine the empirical formula for a compound that contains 1.71 g C and 0.573 g of H. OA. C3H8 O…
A: Given that, Mass of carbon = 1.71 gMass of Hydrogen = 0.573 gMolar mass carbon = 12 g/molMolar mass…
Q: An unknown compound X has the molecular formula C6H140. Compound at 3000 cm. The 'H NMR spectral…
A:
Q: A) OH B) C) D) F Br
A:
Q: 6. Propose a mechanism for the following transformation: Et Me H OH OH Et Me [H2SO4]
A: Given that, the reaction is:
Q: Two monosaccharide isomers with 4 chiral carbons in which the configuration of all of chiral carbon…
A: Answer:When all 4 atoms or groups bonded to a carbon atom are different then such carbon is called…
Q: 13. Use the equation below to answer the following questions: 2Ag(NO3)2(aq) + Cu (s) → 2 Ag (s) +…
A: Oxidation is defined as an increase in the oxidation state of the element.It can also be defined as…
Q: Question Content Area The reaction of NO2 (g) and CO (g) is thought to occur in two steps: Step 1…
A: Overall stoichiometric reaction: NO2(g) + CO(g) = NO(g) + CO2(g) Explanation:Step 1: The…
Q: 8. Ethers can often be prepared by SN2 reaction of alkoxide ions, RO, with alkyl halides. Suppose…
A: The objective of the question is to determine the best route to prepare cyclohexylmethylether using…
Q: The following initial rate data are for the reaction of UO2 + with hydrogen ion in aqueous…
A: Rate=k[UO2+]2[H+]k=156 M−2⋅s−1Explanation:
Q: Explain using equations the process by which you determined the order of stability in Step 23. (Put…
A: The stability of the complex ion can be calculated using the formation constant of the complex ion.…
Q: How many benzene derivatives with molecular formula Совгагн Face are possible? 214575 a) 1 b) 2 3 4…
A: Using the molecular formula we will try to draw different derivatives. We have to keep in mind, the…
Q: Gifying Functional Groups Identify the appropriate group of atoms by selecting each atom…
A: We are given an organic molecule and we have to predict the functional group present in the given…
Q: Write balanced half-reactions for the following redox reaction: 2 NO3(aq) + 2H2O(l)+21¯ (aq) →…
A: Given redox reaction,Since,Redox reaction are those reaction in which oxidation as well as reduction…
Q: Using data from a table of thermodynamic data at 1 atm and 25°C, calculate the equilibrium cor Kp,…
A: Pressure = 1 atm Temperature, T = 25°C = 298 K We have to find Kp
Q: The Cr3+ concentration in a saturated solution of chromium(III) hydroxide is measured and found to…
A: Given,The concentration of Cr3+ = 1.28 x 10-8 M
Q: Which one is aromatic one bond Ile) one compte
A: Conditions for aromaticityCyclic structurePlanar structure should form ring current Follow huckel…
Q: Draw two different Lewis structures, corresponding to the Lewis structures draw two different…
A: We have to draw the Lewis structures, condensed structures and line-angle structures of the given…
Q: Select the group of the periodic table containing some elements with a condensed electron…
A:
Q: Which of the following is an example of a chemical property? O A. Boiling of water O B. Sugar is…
A: Physical property - .No new products are formed, only the physical state of the substance…
Q: Draw the product(s) of the reaction below. Be sure to include lone pairs and single electrons when…
A: Reaction mechanism give the details of a reaction that how it proceeds. Half arrow means transfer of…
Q: NO2
A: Conversion of Aniline to 2,4,6-trichlorobenzamide is given below;
Q: 10. Explain the function of a salt bridge in electrolysis.
A: Given:Determine the function of salt bridge in electrolysis.
Q: A sample of steel (0.1005 g) was dissolved in acid and the solution made up to 100 cm³ with…
A: %Cr (w/w)=7.0607%Explanation:The weight percentage of Cr can be calculated by using the…
Q: Determine the Ka for the acid HA given that the equilibrium concentrations are [HA] = 3.89 M. [A] =…
A: For the given weak acid, , the equilibrium concentrations are,Equilibrium constant of the given acid…
Q: Question 3 Write the mechanism for the following reaction and identify the major product if the…
A: Given that, the reaction is:
Q: Molecule and its Lewis Structure Steric Number, VSEPR Geometry, and Hybridization Bond Length and…
A:
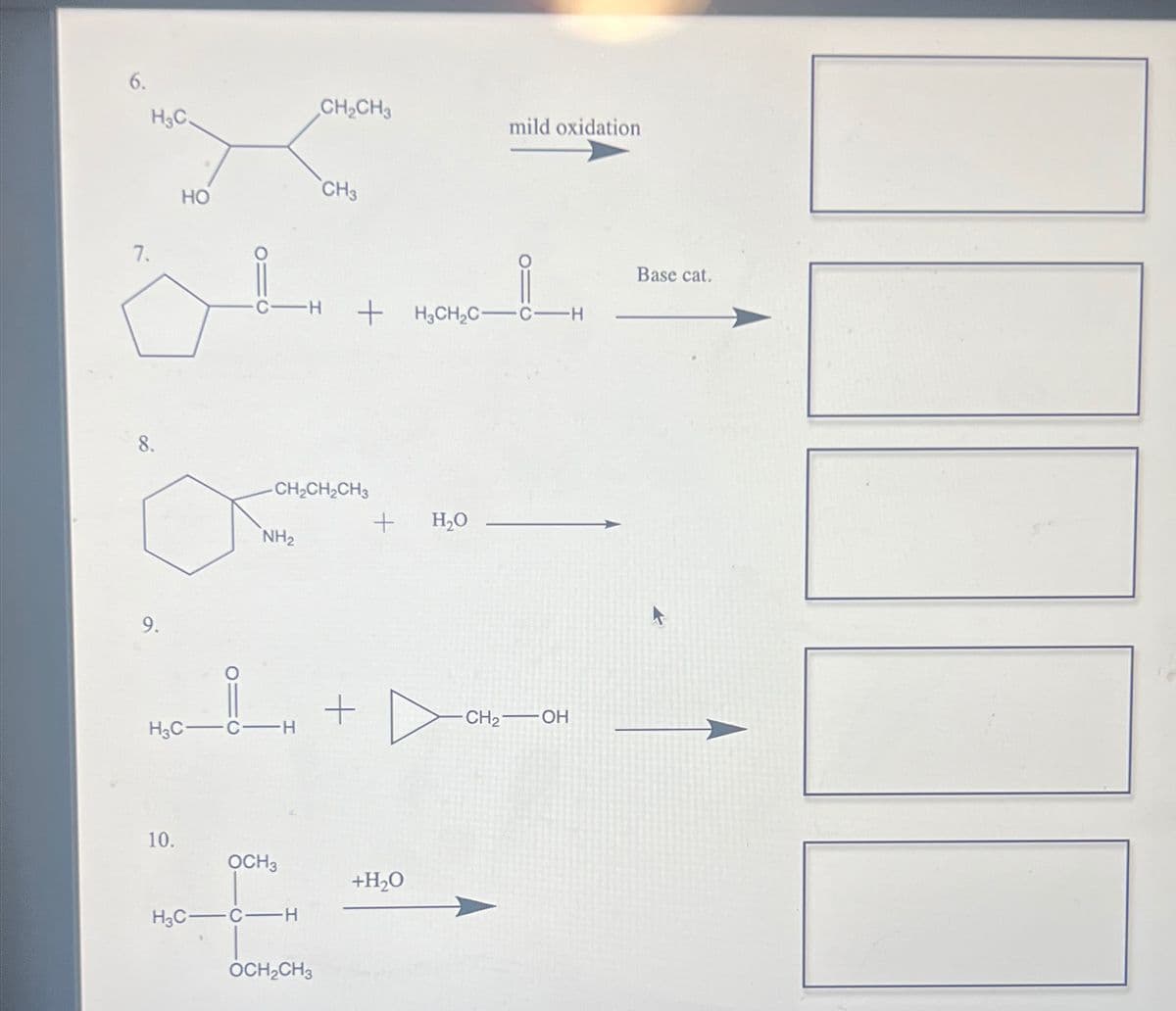
Complete the following reactions mild oxidation 8.9.
Step by step
Solved in 4 steps with 2 images

- What is the concentration of free Hg2+ in 2.6061e-4 M Hg(NO3)2 and 1.1005 M NH3? Hg2+ + 4 NH3 → [Hg(NH3)4]2+ Kf = 1.800e+19Sulfate reduction 5C2H4O2 (acetate) + 10H2O = 10CO2(g) + 40e- +40H+ E0’= (-0.28)*(-1) 4SO4- + 40e- + 40H+ = 4H2S(g) + 16H2O E0’= (-0.22) 5C2H4O2 + 4SO4 = 4H2S + 10CO2 + 6H2O full reaction= 0.06 How is 4SO4- + 40e- + 40H+ = 4H2S(g) + 16H2O E0’= (-0.22)? I don't get how it is -0.22 based off the redox tower.Hydrogen sulfide (H2S) is an odorous gas that can be stripped from solution by bubbling air through the solution. H2S when dissolved in water is also a weak acid that can deprotonate to bisulfide ions which are not removed by air stripping. That reaction is ?2?--> H^+ + ??^- With equilibrium constant Ka=0.86x10-7.Find the fraction of hydrogen sulfide in the H2S form at pH 6 and pH 8. At which pH would you expect to have an easier time to strip away H2S gas?
- What is the [CH3CH2CO2−] in a solution which is 0.1416 M in CH3CH2CO2H and 0.1929 M in HNO3? CH3CH2CO2H(aq) + H2O(l) → CH3CH2CO2−(aq) + H3O+(aq) Ka = 1.3000e-5If 33.82 ml of an hno3 solution of unknown requires 29.95 ml of .100m Noah solution to be neutralized what is the concentration of the hmo3 solitionThe Tris buffer system is commonly used in biochemistry as its pKa of 8.1 allows it to buffer close to physiological pH. (CH2OH)3CNH2 + H+ Û (CH2OH)3CNH3+ What concentrations of Tris and Tris H+ are obtained in a 200 mL solution at pH 8.20 containing 6.1g of Tris? The molecular mass of Tris is 121.1 gmol-1 Please do this step by step thank you!
- Calculate the value of the equilibrium constant for each of the following reactions in aqueous solution. a) HC2H3O2 + OH- <--> C2H3O2- + H2O b) C2H3O2- + H+ <--> HC2H3O2 c)HCl + NaOH <--> NaCl + H2O and I need an actual value for Kc not just the Kc formula which I already know. thanks.Which is the principle species in EDTA solution at pH 7? H2Y2- H4Y H3Y- HY3- Y4-when 2.0 of NaOHCO3 was heated the weight of the reside product after heating was 1.0g calculate the percent weight loss on heating
- In extraction of caffeine from tea, CaCO3 (pKa of HCO3- is 10.3) is used to convert tannin to glucose and calcium salt of gallic acid. Explain why glucose is not in its salt form but gallic acid is. Assuming pKa of -OH of glucose is 12, and pKa of -OH of gallic acid is 9.5 and pKa of -COOH of gallic acid is 4.5.What is the concentration of Mn2+ in a solution with an analytical MnY2- concentration of 0.025 M at pH7? Kmny = 6.2 x 1013 a4 = 4.8 x 10-4 A 9.2 × 10-7 M B 8.4 × 10-13 M C 4.0 × 10-16 M D 2.0 × 10-8 MWhat is the identity of the blood red solution formed from C? a. All of these. b. FeSCN+ c. FeNO+ d. FeSCN2+ e. FeNO2+

