Q: Equations A. Zn + H2SO4 > ZNSO4 + H2 B. Mg + 0, - 2MgO C. Na,S + 2HCI 2NACI + H,S D. H,S + S0, - 3S ...
A: If number of atom and charge are balance both side that is products side and reactants side the eq...
Q: 6. Calculate the pH of a solution during a titration of 2.5M HCI with 35mL of 1.25M NaOH A. 12mL of ...
A:
Q: studont performed a titration to detormine the cxact concentracion of NaOHlani. Thec thration was pe...
A: In order to find the 95% confidence interval, we have to calculate the standard deviation using an e...
Q: The addition of O.3800 L of 1.150 M KCI to a solution containing Agt and Pb?+ ions is just enough to...
A: As per the bartleby guidelines we solve the first question for you. Please find the below attachment...
Q: H H. CH3 H-ö-H CH3 -H- H- + H30 ..+ H-O H CH3 H. CH3 H. H H.
A: It is unimolecular elimination reaction (E1) .
Q: How is Na attached to the other atoms in NaHCO3 (sodium bicarbonate)? Shared electrons Opposite char...
A: For the completion of the octet, the element can share or exchange the electrons. By sharing of elec...
Q: According to your calculations, an experiment in lab should have produced 29.95 grams of product. If...
A:
Q: Calculate the [H 3 O+ ] of the solution with pH = 8.15 .
A:
Q: 1. Explain how water's phase diagram differs from those of most substances. What property of water c...
A:
Q: TRUE OR FALSE: Group 14 elements are gases at room temperature and pressure
A: Carbon silicon germanium tin lead are group 14 elements
Q: After combustion with excess oxygen, a 12.501 g of a petroleum compound produced 38.196 g of carbon ...
A:
Q: Consider the formula given below. C4H8 This following represents the molecular formula of an: A) ...
A: ANS. option A) Alkene
Q: Lead (II) nitrate is reacted with sodium iodide. Calculate the number of Pb2+ ions required to form ...
A: Given, Lead (II) nitrate is reacted with sodium iodide Calculate the number of Pb2+ ions required ...
Q: NacIO3 NaCl + O2 ---> When you complete and balance the specific equation, give the following inform...
A: Chemical reaction can written equation form In chemical equation left side reactants , right sid...
Q: In converting from the pH of a solution to the corresponding H3O+ concentration, the following equat...
A: The answer of the given question is given below
Q: Compounds are composed two or more elements chemically combined while mixture are composed of substa...
A: A question based on introduction to chemistry that is to be accomplished.
Q: 4
A: In the given reaction 1-methylcyclohexene converted to 1-iodo-1-methylcyclohexane.
Q: 7. Explain, in terms of electron configuration, why sulfur atoms and oxygen atoms form compounds wit...
A:
Q: Which compound produces three ions per formula unit by dissociation when dissolved in water? aluminu...
A:
Q: In a series of experiments on the decomposition of dinitrogren pentoxide, N2O5, rate constants were ...
A:
Q: +] P. QUESTION 25 Which is true regarding the C-F bond in the compound shown below it is ionic O it ...
A: Answer: In a covalent bond, atoms form the bond by sharing electrons. For a single covalent bond, ea...
Q: A solution was prepared by dissolving 11.0 g of compound in 270.0 g of ethanol raising the boiling p...
A:
Q: c. Indicate the new C-C bonds in each molecule and then provide the organohalides that were used to ...
A:
Q: A chemical engineer is studying the following reaction: HCH;CO,(aq)+CH,NH,(aq) → CH;CO,(aq)+CH,NH,(2...
A:
Q: synthesize these product based on the precursor given below OH 1 from alkene 2 2-methoxyhexane from ...
A: The synthesis are as follows:
Q: An aqueous solution containing 4.87 g of an optically-pure compound was diluted to 8.00 × 10² mL wit...
A: Specific rotation = observed rotation/(concentration × path length) Observed rotation = -2.47° Mass ...
Q: Phosphorus trichloride is produced from the reaction of Phosphorus (P.) and chlorine. P4 C2 - PCI: +...
A: Given reaction --- P4 + Cl2 ----> PCl3 First of all balanced the reaction then after calculate ...
Q: Write the skeletal structures of propanal, acetone and cyclohexanone. What is the major intermolecul...
A: Forces that hold atoms together within a molecule. Intermolecular forces are forces that exist betwe...
Q: Assuming the density of a 5% acetic acid by mass solution is 1.0 g/mL, determine the volume of the a...
A:
Q: Please explain how the following hair styling products provide color to the hair Oxidizing Hair Dye...
A: Please find your solution below : Hair colouring or hair dyeing is a process of changing the hair co...
Q: 12. a. What is the molarity of a solution made by mixing 125 mL of 5.00 M H3PO4 with 325 mL H2O? b. ...
A:
Q: Enthalpy of formation (kJ/mol) Substance A(s) -5.38 A(g) 20.43 A(I) -3.20 B(s) -18.26 B(g) 27.53 C(s...
A:
Q: Atomic Weights of the following elements: N = 14 amu Na = 23 amu C = 12 amu H = 1 amu O = 16 amu 1. ...
A: Since you have posted multiple questions, we'll answer the first one. If you want any other question...
Q: 5. Aspirin СООН Classification: Salicylic acid + acetic acid COOH Reaction: LOCOCH3 HO + CH;COOH Enz...
A: Aspirin is formed by the reaction of salicylic acid and acetic anhydride in the presence of acetic a...
Q: Fill in the blanks: Progress of the reaction This is the reaction diagram of reactant A converted to...
A: Endergonic and exergonic reactions Exergonic reactions are also called spontaneous reactions, becaus...
Q: What is the percent % (w/w), concentration of sucrose in a solution made by dissolving 7.6 g of sucr...
A: Mass of glucose (solute) = 7.6 g Mass of water (solvent) = 83.4 g Percent concentration (weight pe...
Q: Given the following equation: N2 (g) + 3 H2 (g) → 2 NH3 (g) How many grams of H2 are required if 36....
A: Formation of ammonia reaction are given
Q: Use the quadratic formula to calculate the pH of a 0.10M acid solution; K =; 2.3 * 10 ^ - 3 1.85 5.8...
A:
Q: Draw the resulting product(s) from the coupling the given radicals. Inlcude all applicable electrons...
A:
Q: Iron and water react to form iron(III) oxide and hydrogen, like this: 2 Fe(s)+3 H,0(g)→Fe,O3(s)+3 H2...
A:
Q: If 45 mL of water are added to 250. mL of a 0.75 M K2SO4 solution, what will the molarity of the dil...
A:
Q: -. Draw a Moseley plot for the L. X-rays using the following energies in ke V: Mn 0.721 Rh 2.89 Zn 1...
A: I've solved for K beta value of spectral line having certain a and b value. You do the same with the...
Q: OH F F F
A:
Q: Under certain conditions, mixing furan with maleic anhydride in diethyl ether yields a crystalline s...
A:
Q: Tên equations are listed below, Some are synthesis reactions, Some are not. Make a check () in the c...
A:
Q: H2NNH2 CH3 H* H3C `CH3 H3C IZ
A: We have find out mechanism for 3,5 dimethyl pyrazole
Q: 5. What mass of C2H6O is needed to react with 256 g O2?
A: When C2H6O reacts with Oxygen it generates CO2 and H2O. Using stoichiometric of balanced reaction we...
Q: :O: HICIH bent QUESTION 18 What is the geometry relative to the C atom on the left in the Lewis stru...
A: Here we have to determine the molecular geometry of the indicated carbon atom present in organic com...
Q: What is the total number of oxygen atoms on the right-hand side of this chemical equation? 2 Caz (PO...
A: Given :- 2Ca2(PO4)2(s) + 4SiO2(s) + 12C(s) → 4CaSiO3(s) + P4(s) + 12CO(g) To determine :- total num...
Q: In the reaction between 108.21 grams of zinc and 2.92 moles of HCI, what is the maximum mass in gram...
A: Mass of Zinc = 108.21 gram Moles of HCl = 2.92 moles Reaction is., Zn(s) + 2HCl(aq) ---> H2 (g) +...
Step by step
Solved in 2 steps with 1 images

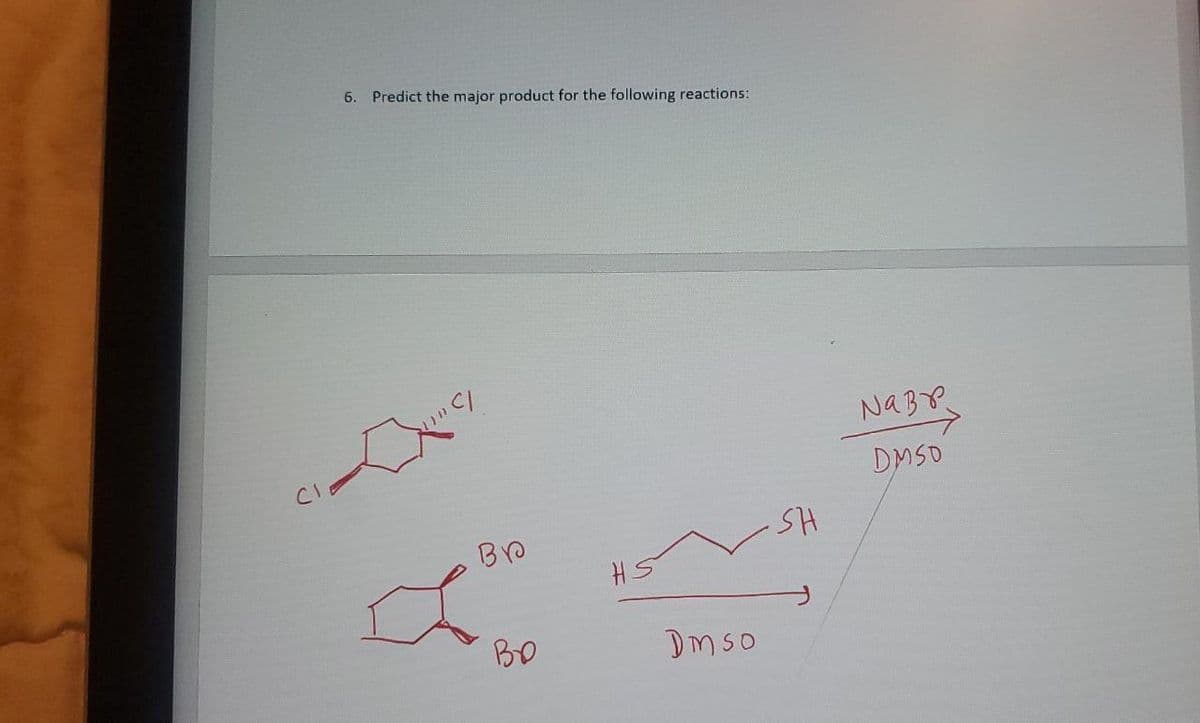
- 1) The Predict the product(s). 2)Label any kinetic/thermodynamic product(s). 3)Give reason towards the selection of kinetic product(s).Orgonic Chmeistry II: 2,4,6-Trinitrotoluene (TNT) is synthesized by trinitrating toluene. The first nitration proceeds much faster than the second two. Briefly explain.9 Predict the major products (including stereochemistry) when cis-3-methylcyclohexanol reacts with the following reagents. SOCl2
- Show each possible product for the reaction below and predict the major product. Be sure to include all geometric isomers. B) Draw the conformer that leads to the major product for the reaction above as a Newman projection. Show the mechanism for its formation. C) How would you change the reaction conditions to get the Hoffman product? include all parts plsArrange the alkyl halides in order of increasing reactivity in an SN2 reaction with KI in acetone (least first). I, IV, III, II II, III, I, IV IV, I, III, II III, II, IV, IWhat major product is expected from the reaction shown in Image 18?

