6.35) Biochemical literature often uses an alternative chemical equation format. For example, the reaction: A + B C + D is often written as: باد B A C D There are eight chemical reactions that occur in the citric acid cycle process. The reactions of the citric acid cycle are shown in the figure on the right. Which reaction is a hydration reaction? HO-CH Reaction 7 CH II CH =O malate T CH₂ O fumarate FADH₂ Reaction 6 H+ + NADH FAD NAD+ жно -H₂O =O Reaction 8 CH₂ CH, O succinate o GTP 0=C CH₂ O oxaloacetate H-CoA acetyl-coenzyme A Reaction 5 GDP CH-C-CoA P₁ H₂O Reaction 1 Reactions of the Citric Acid Cycle =0 I CH₂, CH₂ H-COA 0=C H+ -0₂ CH₂ HO-C- CO, 10 CH₂ Reaction 4 - 0 H-COA Fo Reaction 2 citrate o Reaction 3 CO, CH₂ CH₂ 0=C NAD+ a-ketoglutarate =0 HỌ–CH CH₂ CH- isocitrate C=O NAD+ H+ + NADH
6.35) Biochemical literature often uses an alternative chemical equation format. For example, the reaction: A + B C + D is often written as: باد B A C D There are eight chemical reactions that occur in the citric acid cycle process. The reactions of the citric acid cycle are shown in the figure on the right. Which reaction is a hydration reaction? HO-CH Reaction 7 CH II CH =O malate T CH₂ O fumarate FADH₂ Reaction 6 H+ + NADH FAD NAD+ жно -H₂O =O Reaction 8 CH₂ CH, O succinate o GTP 0=C CH₂ O oxaloacetate H-CoA acetyl-coenzyme A Reaction 5 GDP CH-C-CoA P₁ H₂O Reaction 1 Reactions of the Citric Acid Cycle =0 I CH₂, CH₂ H-COA 0=C H+ -0₂ CH₂ HO-C- CO, 10 CH₂ Reaction 4 - 0 H-COA Fo Reaction 2 citrate o Reaction 3 CO, CH₂ CH₂ 0=C NAD+ a-ketoglutarate =0 HỌ–CH CH₂ CH- isocitrate C=O NAD+ H+ + NADH
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter8: Thermochemistry
Section: Chapter Questions
Problem 23QAP: In the late eighteenth century Priestley prepared ammonia by reacting HNO3(g) with hydrogen gas. The...
Related questions
Question
Answer is Reaction 7? Can you explain to me, please?
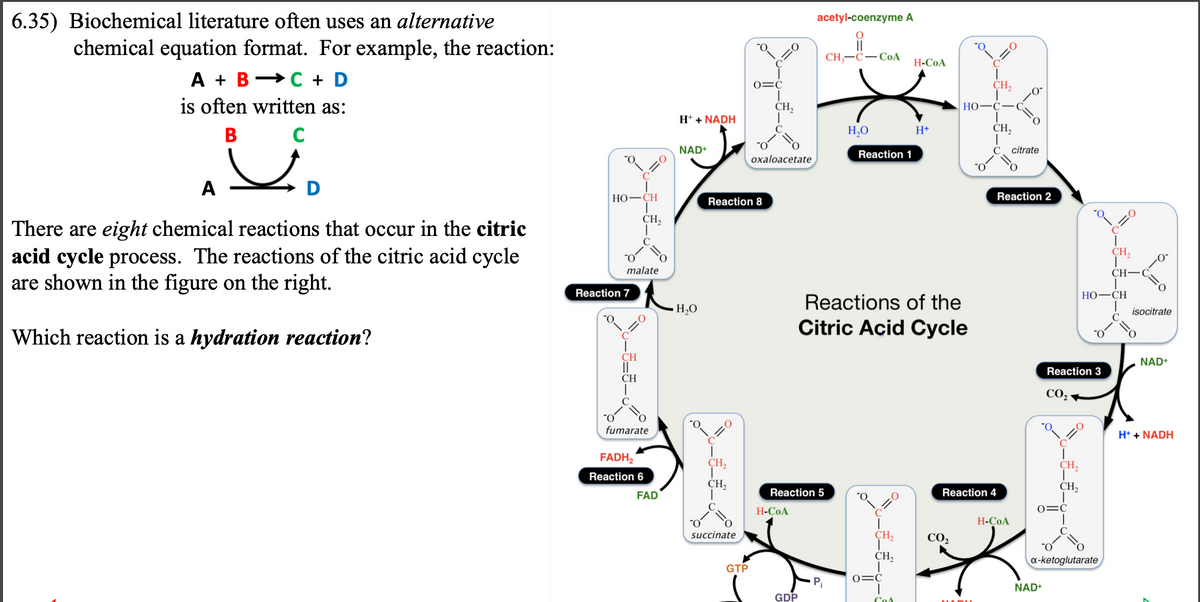
Transcribed Image Text:6.35) Biochemical literature often uses an alternative
chemical equation format. For example, the reaction:
A B C + D
is often written as:
B
A
There are eight chemical reactions that occur in the citric
acid cycle process. The reactions of the citric acid cycle
are shown in the figure on the right.
Which reaction is a hydration reaction?
D
*O₂
HO–CH
(=O
Reaction 7
malate
CH
||
CH
CH,
大
fumarate
FADH₂
Reaction 6
FAD
H+ + NADH
NAD+
H₂O
CH₂
CH₂
succinate
"O
Reaction 8
GTP
0=C
CH₂
oxaloacetate
H-COA
acetyl-coenzyme A
O
||
CH₂-C-CoA
Reaction 5
GDP
H₂O
Reaction 1
"O
Reactions of the
Citric Acid Cycle
C
:0
CH₂
CH₂
O=C
P₁
H-COA
Cos
H+
HO
CO₂
Reaction 4
JADU
=0
CH₂
CH₂
O
H-COA
citrate
Reaction 2
Reaction 3
CO₂
CH₂
CH₂
O=C
NAD+
HO–CH
x-ketoglutarate
CH,
T
CH
O
isocitrate
NAD+
H+ + NADH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning