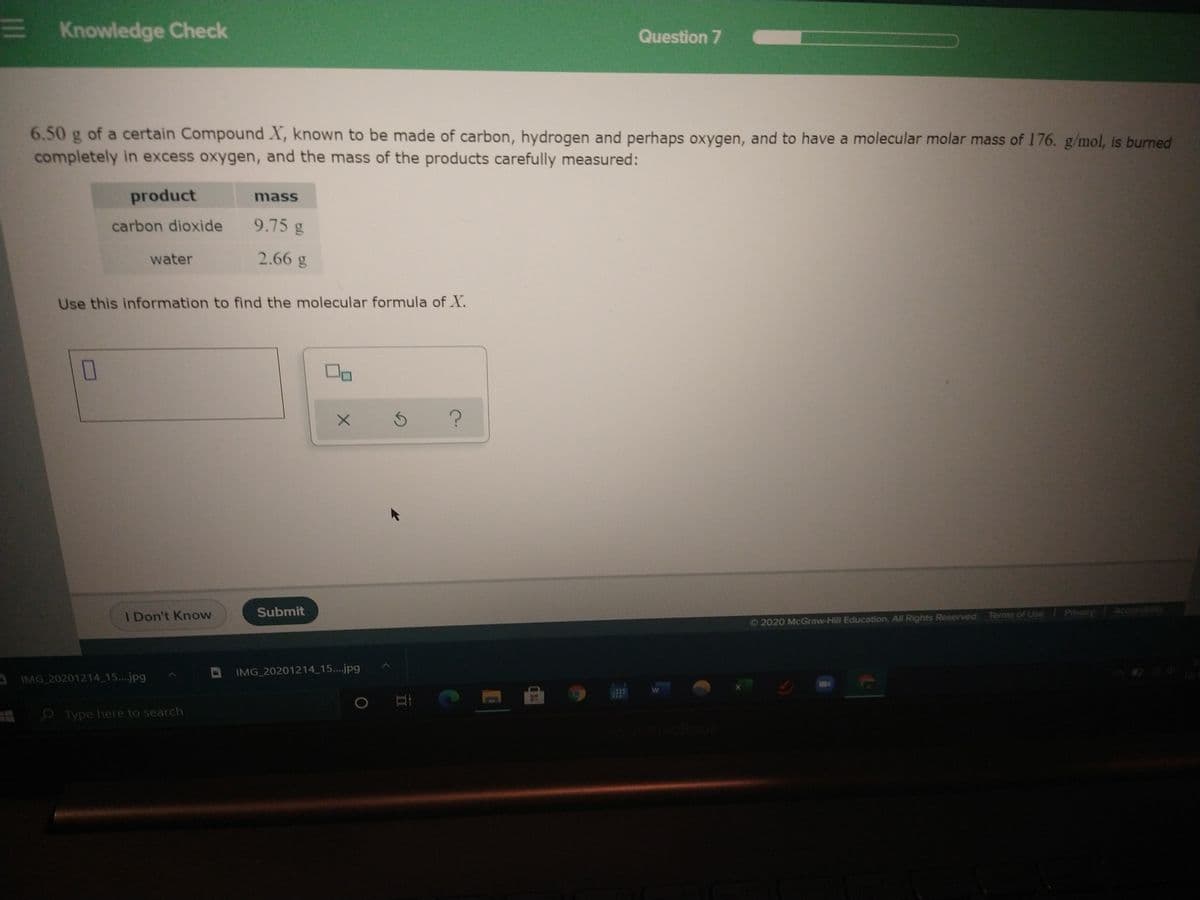
6.50 g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have a molecular molar mass of 176. g/mol, is burned completely in excess oxygen, and the mass of the products carefully measured: product mass carbon dioxide 9.75 g water 2.66 g Use this information to find the molecular formula of X.
6.50 g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have a molecular molar mass of 176. g/mol, is burned completely in excess oxygen, and the mass of the products carefully measured: product mass carbon dioxide 9.75 g water 2.66 g Use this information to find the molecular formula of X.
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Chapter3: Calculations With Chemical Formulas And Equaitons
Section3.2: The Mole Concept
Problem 3.1CC: You have 1.5 moles of tricycles. a How many moles of seals do you have? b How many moles of tires do...
Related questions
Question
Transcribed Image Text:E Knowledge Check
Question 7
6.50 g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have a molecular molar mass of 176. g/mol, is burned
completely in excess oxygen, and the mass of the products carefully measured:
product
mass
carbon dioxide
9.75 g
water
2.66 g
Use this information to find the molecular formula of X.
I Don't Know
Submit
Privacy
Accessibity
O2020 McGraw-Hill Education, All Rights Reserved. Terms of Use
DIMG 20201214 15..jpg
IMG 20201214 15...jpg
O Type here to search
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning