Chapter16: Electromagnetic Waves
Section: Chapter Questions
Problem 101AP: If a Lightsail spacecraft were sent on a Mars mission, by si1at ratio of the final force to the...
Related questions
Question
100%
Transcribed Image Text:Design
Transitions
Animations
Slide Show
Review
View
O Tell me
Layout v
A A A
Ó Shapes
Calibri (Body)
12
Reset
IA v 3 v Convert to
SmartArt
Arrange Qulck
Styles
Design
Ideas
Picture
A Text Box
Sensitivity
В
I U
ab
x'
x, AV - Aa v
Section v
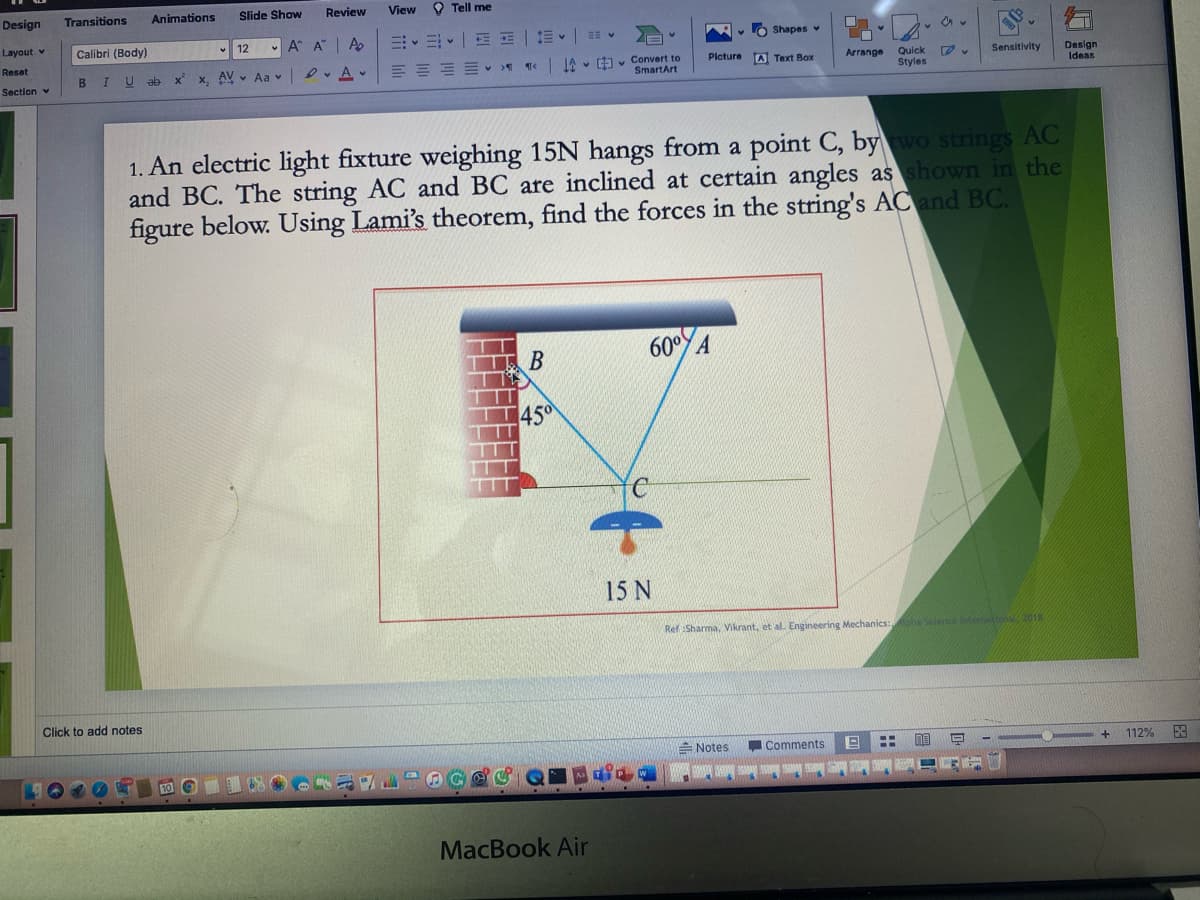
1. An electric light fixture weighing 15N hangs from a point C, bywo strings AC
and BC. The string AC and BC are inclined at certain angles as shown in the
figure below. Using Lami's theorem, find the forces in the string's ACand BC.
60° A
В
1450
15 N
Ref Sharma, Vikrant, et al. Engineering Mechanics: h Sciece Intema , 2018
Click to add notes
E Notes
Comments
::
112%
MacBook Air
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

