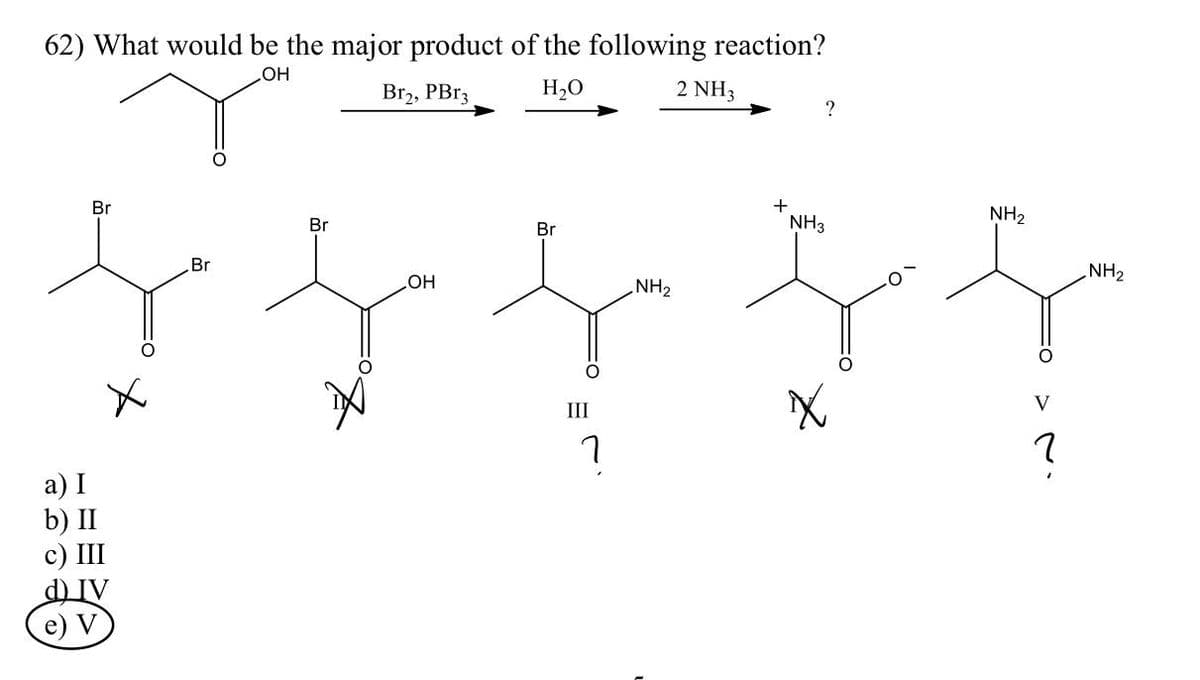
62) What would be the major product of the following reaction? OH Br2, PBr3 H₂O 2 NH3 Br a) I ) III DIV e) V Br Br OH Br III า NH₂ + ? NH3 X NH₂ V ? NH₂
Q: Suppose you discovered a new amino acid. Its R-group contains only hydrogen and carbon atoms.…
A: Hydrophobic and hydrophilic are terms used to describe the interactions between substances and…
Q: Draw structures for the alkene (or alkenes) that gives the following reaction product. CH3 CH3 I I…
A:
Q: If 8.5 mols of A and 11.5 mols of B react according to the reaction below how much energy will be…
A: Since the enthalpy change of the reaction is positive the reaction is endothermic and hence energy…
Q: Complete the following reaction by drawing the structure of the major product(s) expected. CH₂…
A: Alkene gives an electrophilic addition reaction with hydrogen halide to form an alkyl halide. The…
Q: Which statement about reaction shown below is CORRECT? H C HCOH CH₂OPO Glyceraldehyde 3-phosphate O…
A: The nicotinamide adenine dinucleotide (NAD+)/reduced NAD+ (NADH) and NADP+/reduced NADP+ (NADPH)…
Q: Draw a possible retrosynthesis for this product structure with specific reagents. O Net ₂
A: The given compound to made is We have a benzene ring to which an amino group, an ester group as…
Q: what is the temperature of 1.50 moles of argon gas that is contained in a 500.0 ML flask at 15.8…
A: Moles of Argon , n = 1.50 mol Volume of flask , V= 500 mL = 500×10-3 L Pressure of flasK , P = 15.8…
Q: Consider a hypothetical chemical reaction between compound A and compound B, which produces compound…
A: In chemical reactions, the presence of an activation energy barrier can hinder the progress of even…
Q: You will find it useful to keep in mind that HF is a weak acid. 0.52 mol of KOH is added to 1.0 L of…
A: Given,moles of KOH =0.52 molMolarity of HF = 1.3 MVolume of solution = 1.0 Lmoles of HBr =0.31…
Q: A. The Lewis diagram for SiCl4 is: :CI: Si-CI: There are the geometry of SiCl4 is :CI: The…
A:
Q: Consider the following data on some weak acids and weak bases: hydrofluoric acid name nitrous acid…
A: We need to arrange the given solutions in order of increasing pH based on the given Ka and Kb…
Q: Draw the alkene that can be used to prepare the following alcohol as the exclusive product of a…
A:
Q: The reaction: H2(g) + I2(g) → 2HI(g) is a second order reaction (first order with respect to each of…
A: Given,The reaction,H2(g) + I2(g) →2 HI(g)at,H2=0.200 mol/LI2=0.200 mol/L The rate -dH2dt = 0.00097…
Q: The ¹H NMR spectrum of the following molecule shows three peaks. Which is the expected ratio of…
A: As we know, the number of signals in the PMR spectrum of an organic compound tells us as to how many…
Q: Identify the element with each of the following electron configurations. (a) [kr]5s¹ (b) [Ne]3s²3p4…
A: The atomic numbers of the noble gases Kr, Ne, and Ar are 36, 10, and 18 respectively.
Q: Shown below is the major resonance structure for a molecule. Draw the second best resonance…
A: Rules for deciding the most stable resonance structures: 1. Uncharged structure is more stable than…
Q: When the following molecular equation is balanced using the smallest possible integer coefficients,…
A: Given reaction: nitrogen (g)+oxygen (g) → nitrogen monoxide (g)
Q: What is the correct product of the following transformation? OA O OC OD OE of A H-B mat Kot 없 B C Få…
A:
Q: Researchers tried to explain how vesicular transport occurs in cells by attempting to assemble the…
A: a) motor proteins Vesicular transport within cells typically occurs with the help of motor proteins,…
Q: If a molecule of DNA contains 10% cytosine, it has adenine, respectively. a) 10%; 40% b) 10%; 80% c)…
A: 11. Given that in a DNA molecule the percentage of cytosine is 10%. We have to calculate the…
Q: Atoms of X decay to atoms of Y with a half-life of 1,000 years. If there are 2,000 atoms of X to…
A:
Q: Draw a structural formula for heptane. • You do not have to consider stereochemistry. • You do not…
A: The organic compounds in which carbon and H atoms are connected via single bonds, are called…
Q: 5. An unbalanced reaction of iron (III) chloride and ammonium hydroxide is shown below. When 50.0 g…
A:
Q: What is the IUPAC name of the following compound? CH3 CH3CCH₂CH₂ CH
A:
Q: Give the IUPAC name of the product that would form when the following cycloalkene undergoes…
A: Catalytic hydrogenation is a process in which an unsaturated compound is converted to saturated…
Q: 4. Sodium metal (Na) reacts with chlorine gas ( I Cl₂) to produce solid sodium chloride (NaCl). (a)…
A: A balanced chemical equation contain equal number of atoms of each elements on both sides of the…
Q: What is the IUPAC name of the following substance? CH3 HC=CCH₂CHCH3
A: The IUPAC Name of the Compound is written in the following format: locant + prefix + root + locant +…
Q: olve correctly please. Can someone draw out the mechanism for this reaction
A: In this reaction conversion of acyl chloride into tertiary alcohol by attack of two moles of gilman…
Q: Why is it not possible to prepare the following carboxylic acid by a malonic ester synthesis? Select…
A: A question based on reactions of ester. 4 options for the reason for the failure of the reaction are…
Q: Draw a Fischer Projection formula for the enantiomer of each of the following monosaccharides.
A: Enantiomers are non-superimposable mirror images of each other.
Q: A compound contains only C, H and O. The mass spectrum shows peaks at m/z = 58 and 43. What is the…
A: Mass spectrum is the technique used in analytical chemistry for the separation of ions based on the…
Q: raw the structure and look up the boiling points for the four compounds listed below.…
A: ->The boiling point is the temperature at which a liquid changes into a vapor. -> As the…
Q: Which plot below best represents the relationship between pressure (P) and volume (V) for an ideal…
A: Given, The ideal gas equation: P V = n R T P→ Pressure V→ volume n→ moles of gas R→gas constant…
Q: Construct the expression for Kc for the following reaction. CO(g) + 2 H,(g) CH₂OH(g) Drag the tiles…
A: For a reaction aA (g) + bB(s) <------> cC(g) + dD(g), the equilibrium constant is given by…
Q: 2. Calculate the cell potential for the cell below at 25 °C Ti(s) Ti(NO3)2(aq) (0.92 M)|| HgCl₂(aq)…
A:
Q: Write a complete balanced equation using structural formulas to show how to fully saturate ethyne…
A: The reaction of ethyne with chlorine involves electrophilic addition.
Q: Question 3 3.1 Convert each Haworth projection below to its acyclic form CH2OH 3.2 a) ОН -І Н Н Н ОН…
A: Haworth projection can be converted in open chain structure:
Q: HN N=
A: The oxidation number represents the loss or gain of electrons to the respective elements of the…
Q: If a molecule of DNA contains 10% cytosine, it has adenine, respectively. a) 10%; 40% b) 10%; 80% c)…
A: 12) we have to calculate the percentage of guanine and adenine 13) we have to explain why is DNA…
Q: You walk 3.67 miles. Your pace is 4 miles per hour. Your stride is 2.6 feet. How many strides will…
A: Dimensional analysis is used to convert a physical quantity given in one type of unit to some other…
Q: Describe the prepartion of 4.00L of 0.175 HClO4,starting with an 8.00M solution reagent.
A:
Q: A peptide bond is a) a particularly unstable covalent bond b) an ionic bond, not a covalent one c) a…
A: Proteins serve essential functions in organisms, playing roles in structural support, chemical…
Q: dentify the relationship between the labelled protons as diastereotopic for the molecules below. on…
A: Homotopic protons - are those whose positions can be interchanged by rotation about the proper axis…
Q: Compare the 13C NMR spectra of isoborneol and camphor. Explain which peaks can be used to determine…
A: 13 C NMR spectroscopy is useful to determine types of carbons, their signals, splitting etc hence we…
Q: Recent technological advances have made it more feasible than ever to work out the three-dimensional…
A: 7) allosteric regulation is a type of regulation where a protein regulate by binding an effect or…
Q: Y Part A Draw structural formulas for all of the possible isomers of n-pentyne that can be formed by…
A: Isomers: compounds having the same molecular formula but different physical and chemical properties.…
Q: Carry out the following conversions. Report your answers to the correct number of significant…
A: To solve this problem we have to do the given unit conversion. Note : Since you have asked…
Q: 13. The chemical equation for the decomposition of potassium chlorate ( 2 KCIO, (s) 2 KCl(s) + 3…
A: For the calculation of the mass of the product we use the following steps: Balance the given…
Q: A solution is made by combining 250 mL of 0.010 M Mg(NO3)2 and 250 mL of 0.100 M NaF. Calculate the…
A:
Q: 1. 2. 3. Write the correct name for the following compound. CH₂-CH3 H₂C-CH₂ H3C-CH-CH-CH LI CH3 CH₂…
A: 1. Select longest carbon chain as parent chain . 2. Start numbering from where we get substituent…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- What is the final product of the following reactions? A) I B) II C) III D) IV2) What is the main product for each of the following reactions? (d) Styrene + CH3CH2COCl AlCl3, then Br2, Fe -----> ? (e) Benzene + 1-chloro-2,2-dimethylpropane, AlCl3 -----> ? (h) Benzene + HCOCl, AlCl3 then 1-chlorobutane, AlCl3 -----> ?possible products of the reaction below
- What would be the majority product of the following reaction?: [Pt(NH3)4]2+ + 2 Cl- → ? Select one:a. No reaction because chloride is not a good inbound binder b. trans-[PtCl2(NH3)2] c. cis-[PtCl2(NH3)2] d. [PtCl(NH3)3]+Predict the product of the following reaction and classify the reaction. Pb(NO3)2+FeSO4--->PbSO4+________3. What is the main product for each of the following reactions? (a) p-Methylanisole + CH3CH2COCl, AlCl3 --------> ? (b) Toluene + Br2, light, then CH3CH2COCl, AlCl3 --------> ? (c) Acetophenone + CH3CH2Cl, AlCl3, then Br2, Fe -----> ? (d) Styrene + CH3CH2COCl AlCl3, then Br2, Fe -----> ? (e) Benzene + 1-chloro-2,2-dimethylpropane, AlCl3 -----> ? (f) Benzene + HCOCl, AlCl3 then 1-chlorobutane, AlCl3 -----> ? 4. Using arrows to show electron movement, write the stepwise mechanism for the major products in the reactions (3a and 3f) listed above.
- What is a possible formula for a carbocation with m/z=97Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) KMnO4, H3O+ CH3Cl, AlCl3 HNO3, H2SO4 Cl2, FeCl3 fuming sulfuric acidPredict the product(s) and provide the mechanism for each reaction below.
- Carvone is the major constituent of spearmint oil. What products would you expect from reaction of carvone with the following reagents? a) (CH3)2CU-Li+, then H3O+ b) LiA1H4, then H3O+ c) CH3NH2 d) C6H5MgBr, then H3O+ e) H2/Pd f) CrO3, H3O+ g) (C6H5)3 P+C- HCH3 h) HOCH2CH2OH, HC16 H+ + 6 I- + ClO3- → 3 I2 + 3 H2O + Cl- What is the oxidizing reagent in this reaction?Rank the species below in order of increasing nucleophilicity in protic solvent. I. H2O II. CH3S— III. CH3COO— IV. t-BuO— I, II, IV, III I, III, II, IV I, III, IV, II I, II, III, IV

