7. What molecule does the cell use as an energy carrier? Draw its structure.
Biology (MindTap Course List)
11th Edition
ISBN:9781337392938
Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Chapter7: Energy And Metabolism
Section: Chapter Questions
Problem 15TYU
Related questions
Question
please help me answer these last three questions please please... even without explanation
7. What molecule does the cell use as an energy carrier? Draw its structure.
8. Why is it that this energy carrier is considered to be high energy containing phosphate?
9. Bond of this energy carrier of cells is broken through what?
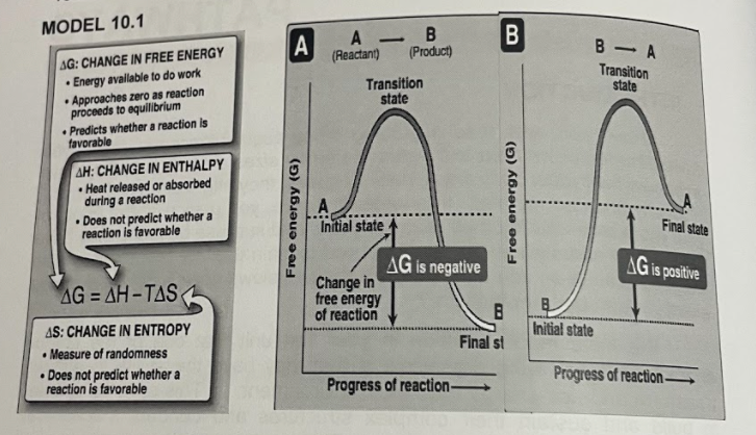
Transcribed Image Text:MODEL 10.1
A
(Reactant)
-
B- A
AG: CHANGE IN FREE ENERGY
Energy available to do work
Approaches zero as reaction
proceeds to equillibrium
• Predicts whether a reaction is
favorable
(Product)
Transition
state
Transition
state
AH: CHANGE IN ENTHALPY
• Heat released or absorbed
during a reaction
• Does not predict whether a
reaction is favorable
A
Initial state
Final state
AG is negative
AG is positive
Change in
free energy
of reaction
AG = AH-TAS
%3D
AS: CHANGE IN ENTROPY
Initial state
Final st
Measure of randomness
• Does not predict whether a
reaction is favorable
Progress of reaction-
Progress of reaction–
B
(5) KBJoue 00
Free energy (G)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax