8) CH3 CH3 CH3 CH3 A Bo H Н H H Answer the following questions, which refer to the two acetonide diastereomers shown above. a) Would you expect any difference in the number of peaks in the ¹H NMR spectra of each acetonide? (Y or N), b) Would you expect any difference in the number of peaks in the 13C NMR spectra of each acetonide? (Y or N).
8) CH3 CH3 CH3 CH3 A Bo H Н H H Answer the following questions, which refer to the two acetonide diastereomers shown above. a) Would you expect any difference in the number of peaks in the ¹H NMR spectra of each acetonide? (Y or N), b) Would you expect any difference in the number of peaks in the 13C NMR spectra of each acetonide? (Y or N).
Chapter14: Conjugated Compounds And Ultraviolet Spectroscopy
Section14.SE: Something Extra
Problem 53AP: Treatment of 3,4-dibromohexane with strong base leads to loss of 2 equivalents of HBr and formation...
Related questions
Question
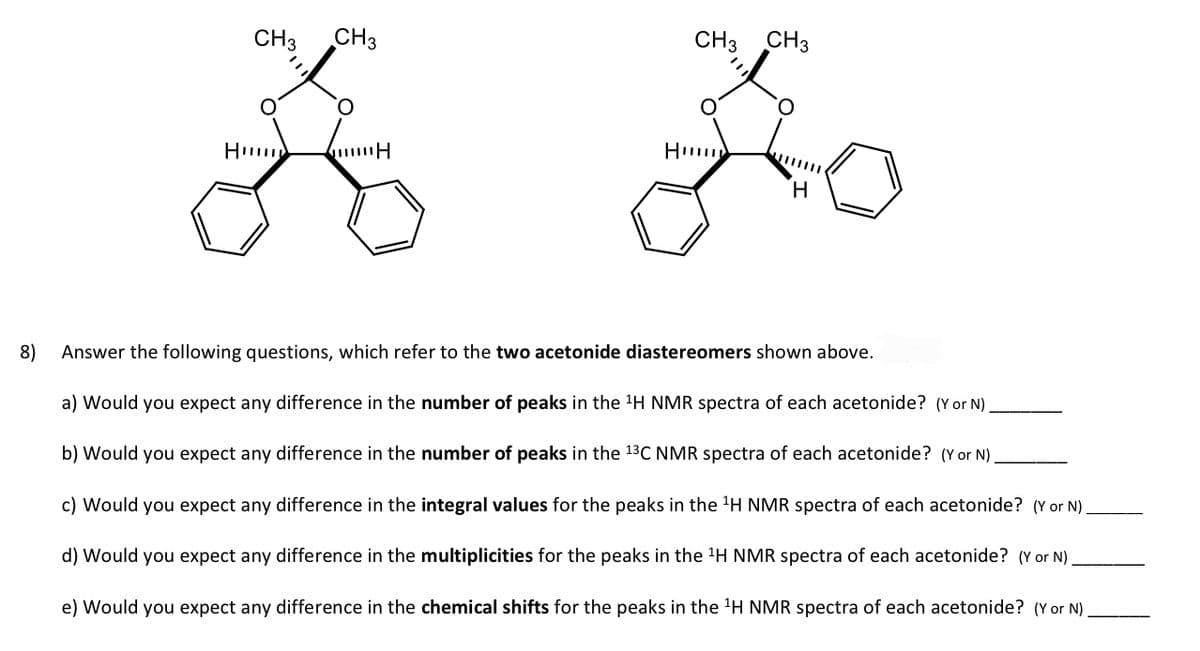
Transcribed Image Text:8)
CH3 CH3
O
36
Hu
H
CH3 CH3
Fo
H
H
Answer the following questions, which refer to the two acetonide diastereomers shown above.
a) Would you expect any difference in the number of peaks in the ¹H NMR spectra of each acetonide? (Y or N).
b) Would you expect any difference in the number of peaks in the 13C NMR spectra of each acetonide? (Y or N)
c) Would you expect any difference in the integral values for the peaks in the ¹H NMR spectra of each acetonide? (Y or N)
d) Would you expect any difference in the multiplicities for the peaks in the ¹H NMR spectra of each acetonide? (Y or N).
e) Would you expect any difference in the chemical shifts for the peaks in the ¹H NMR spectra of each acetonide? (Y or N)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole


Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole