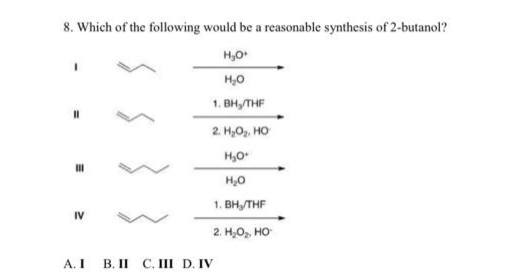
8. Which of the following would be a reasonable synthesis of 2-butanol? НО- НО 1. BHy/THF 2 H,Ộ, HỌ но H₂O 1. BHy/THF 2 H O HO . III IV A.I B.II C.II D.IV
Q: 3. Retrosynthesis: propose starting materials for this array of organic molecules we've encountered…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: A student dissolves 14.1 g of sodium hydroxide (NaOH)in 250. g of water in a well-insulated open…
A: Since the temperature of the water increases after the reaction, hence heat is evolved in the…
Q: Draw The fourth intermediate of the Fischer esterication of 6-hydroxyhexanoic acid.
A: Since the entire reaction is reversible, the Fischer Esterification Mechanism must include either…
Q: Draw the lewis structure with formal charges. If the lewis structure has resonance please show all…
A: Lewis structure is a method of representing bonding pattern in a molecule.It tells us how different…
Q: ? НО
A: Given is organic synthesis reaction.In this reaction alkene is converted into alcohol with extra two…
Q: NBS light
A:
Q: Consider the following multistep synthesis. Br KOtBu What is the identity of 1? 1 O CH3OH O CH3CH₂OH…
A: The desired alkene in the first step can be formed by an E1 reaction along with the rearrangement of…
Q: For each organic compound in the table below, enter the locant of the highlighted side chain. CH3 1…
A: The parent chain is the chain that contains a maximum number of carbon atoms and if there are two…
Q: Question 1 - Which of the following line-bond structures below are representative of the Newman…
A: The objective of the question is to draw the line-bond structure of the given Newmann projection.
Q: Calculate the pH for each case in the titration of 50.0 mL of 0.180 M HClO(aq) with 0.180 M KOH(aq).…
A: The question is based on the concept of the pH of the solution.It is defined as a negative logarithm…
Q: An atom of hellum has a radius e = 31. pm and an average speed in the gas phase at 25°C of 787.m/s.…
A: rHe = 31. pmAverage speed in gas phase = 787.m/s Temperature= 25°C = 298K Uncertainty in speed =…
Q: ОН Br
A: Conversion of alkyl bromide into tertiary alcohol by using grignard reagent
Q: Draw the major monobromination product of this reaction. NBS, light (6)
A: The given reaction is .We have to give the major product of the reaction.
Q: 11. What is the hydrogen ion concentration of a glass of orange juice, with a pH of 2.8? done
A: pH of orange juice = 2.8We need to calculate the hydrogen ion concentration of a glass of orange…
Q: Determine the potential of a platinum electrode when placed in a mixture of 0.0244 M potassium…
A:
Q: List the name and position for each substituent in this organic molecule on a separate row of the…
A: The position of the substituents present in the given compound can be decided by numbering the…
Q: Part A Using the equation E = (hcRH) (-³) = Submit na = (-2.18 x 10-¹8 J) calculate the energy of an…
A: The objective of the question is to calculate the energy of an electron in a hydrogen atom when the…
Q: What is the balanced chemical equation for the chemical reaction depicted in the figure? Be sure…
A: Information of question
Q: 3. Draw the four 2'-deoxyribose 5'-monophosphate nucleotides found in DNA. Label the nucleotides…
A: Nucleotides are composed of A nitrogenous baseA pentose sugarA phosphate groupThere are two types of…
Q: Identify whether each species functions as a Brønsted-Lowry acid or a Brønsted-Lowry base in this…
A: According to Bronsted and Lowry concept,Acid is a substance that has the tendency to donate a…
Q: OTBS I !!! OH Me™! Im Me H OMe OMe OTIPS Br OTIPS tBuLi, Et₂O, -78 °C 83% OTBS I Me Me I !!!! "H OMe…
A: Carbonyl C atom is electron-deficient C atom. Hence, it is electrophilic in nature. An electrophile…
Q: If the system 2CO(g) + O2(g) → 2CO2(g) has come to equilibrium and then more CO(g) is added. What…
A:
Q: Which is the proper IUPAC name of the major organic product of the reaction provided? A B OC Cl₂, hv…
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: Consider the following equation: KOH(s) + CO₂(g) → K₂CO₃(s) + H₂O(l) Calculate the mass in grams of…
A:
Q: Draw the Lewis structure (with formal charges) of the indicated chemical species. If there is…
A: Lewis’s bonding theory is based on the octet rule. The Lewis structure is a simplified…
Q: Suggest typical reagents needed to convert an alkene into an aldehyde (e.g. B to C). Your answer may…
A: Alkenes react with ozone to cleave the double bond oxidatively by forming ozonides. The ozonide…
Q: What is the molar solubility of AgBr in 0.180 M NaBr? 0.09 M 8.0 x 10-11 M 4.2 × 10-¹2 M
A: Information of question
Q: Using the Born-Haber cycle, the AHf of KBr is equal to - As used here AH₁°[K(g)]=AHsub, and AH…
A: From the Born-Haber cycle, one can calculate the lattice enthalpy of an ionic crystal. It involves a…
Q: Suppose you were preparing 1.0 L of a bleaching solution in a volumetric flask, and it calls for…
A: Answer :volume = mole* 1000/ concentrationvolume of stock solution = (0.24*1000)/0.70 = 342.85 mL
Q: Draw complete reaction mechanism for this reaction and lable to product.
A: Carbonyl compounds (aldehyde and ketone) react with an alcohol to form hemiacetal in the presence of…
Q: An unknown compound contains only C, H, O. Complete combustion of 5.50 g of this compound produced…
A:
Q: C. b C a CN d
A:
Q: Draw the BEST lewis structure (with formal charges) of the indicated chemical species. (Draw all…
A: Lewis’s bonding theory is based on the octet rule. The Lewis structure is a simplified…
Q: When a 21.0 mL sample of a 0.342 M aqueous hydrocyanic acid solution is titrated with a 0.495 M…
A:
Q: where did the x 10-6 come from on the 12.8 concentration value when it wasn't in the original…
A: Question :Calculate the extinction coefficient of each peak if the measurements were made for a…
Q: A research student used a new software package to calibrate the electronic map using a material…
A: The objective of the question is to determine which atom, gallium (Ga) or bromine (Br), would have…
Q: The compound below can be prepared with an alkyl iodide and a suitable nucleophile. Identify the…
A:
Q: Calculate the freezing point of each of the following solutions (Ftemperature changes for 1 mole of…
A:
Q: When the ammonium salt CH,CH₂CH, NHBr is treated with NaOH, the products are H₂0, NaBr, and an…
A: Tertiary ammonium bromide react NaOH to give tertiary amine , water and NaBr
Q: Draw two electron-dot resonance structures that obey the octet rule for trichloronitromethane,…
A: Lewis’s bonding theory is based on the octet rule. The Lewis structure is a simplified…
Q: the lewis structure with formal charges. If the lewis structure has resonance please show all…
A: Here we have to calculate Lewis structure for SeO2 O4 - and its resonance structure if available.
Q: TRANSMITTANCEIL LOD D 4000 Please design a synthesis to make the following compound from ethane.…
A: The question is based on the concept of organic spectroscopy. We need to analyse the spectral data…
Q: Two sets of ionizations are shown in the tables below. Complete the tables by ordering each set of…
A: Answer:Ionisation energy is the amount of energy required to remove the electron from an isolated…
Q: 4. Write a mechanism that explains formation of the products shown in the following reaction. Br2,…
A: The addition of bromine to an alkene in a nonpolar solvent produces vicinal dibromo alkane.
Q: Give the name of the following molecule. S.
A: The alkyl sulfanyl group with shorter carbon chain is a substituent.The longest carbon chain is the…
Q: d. NH₂ i i 2) LIAIH4, then H₂O workup
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Consider an aqueous NaOH solution that contains 20 wt% NaOH and 80 wt% H₂O (MWNaOH = 40, MWH20 =…
A: Note: Since you have posted a question with multiple sub-parts, we will provide the solution only to…
Q: An evacuated reaction vessel is filled with 3.60 atm of NOCI. When the system reaches equilibrium…
A: We need to calculate Kp for this reaction: Given:Initial pressure of NOCl = 3.60 atmAt equilibrium,…
Q: how would you answer this question? this is a non graded practice
A: The objective of the question is to understand the effect of a molecule absorbing a photon of…
Q: Pentaborane-9 (B, H,) is a colorless, highly reactive liquid that will burst into flames when…
A: Answer:Here, are the stoichiometric coefficients of products and reactants respectively.
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- 1. What type of reaction explains solubility of phenol in sodium hydroxide? Explain the organic product formed. 2. How do the reactions of phenol samples with FeCl3 compare? Which structural component of the phenols account for the observationIn the reaction from compound 10 to compound 11, why the C=C bond is retained and is not hydrogenated? f) LiAlH4, Et2O, 08C; g) Ac2O, py, DMAP, CH2Cl2, RT, 77% over 2 steps;Complete the reaction: 42K19 → + β + [a]_____________
- Fill in necessary products reactants or reagants of these reactions. Please note the existence of enantionmers in some cases.The reduction of 4- nitrobenzaldehyde using sodium borohydride,MaBH4 in ethanolwhich is not a characteristic of reduction? liss of oxygen gain of electrons gain of hydrogen loss of protons and illustrates the polarity of a carbonyl group?C=OIn the lab for the synthesis of methyl m-nitrobenzoate from methyl benzoate using HNO3 and H2SO4 via EAS, the product is recrystallized to remove impurities using 95% ethanol. Would o-nitrobenzoate and methyl p-nitrobenzoate be considered impurities? If that is so, how come ethanol can remove o-nitrobenzoate and methyl p-nitrobenzoate, but has no effect on m-nitrobenzoate? Thank you.
- a. Give at least three characteristics of dichloromethane that makes it a good extracting solvent for the alkaloid.b. Why is it necessary to remove a stopper from a separatory funnel when the liquid is being drained from it through a stopcock?c. What are emulsions? Why do they form during extractions? How is the formation of an emulsion minimized? How are emulsions removed?What would the % yield of ester be if the reaction were simply allowed to equilibrate (i.e. no products are removed)? C3H7OH+C2H5COOH=C6H12O2+H2O Percent yield of ester was 71.6% weight of product= 20.7868g 19.0mL (0.25mol) of n- propanol 18.5 mL (0.25 mol) of propionic acid At room temperature, the equilibrium constant, Keq is approximately 3.Acid (phosphiruc acid) is catalyzing this dehydration reaction(cyclphexanol to cyclohexene). What does that mean? How many equivalents of acid were needed for the reaction to proceed?
- Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) KMnO4, H3O+ CH3Cl, AlCl3 HNO3, H2SO4 Cl2, FeCl3 fuming sulfuric acidWhere does the 40% and 60% product come from? How to determine this?complete the reaction 68

