Q: Calculate the binding energy (in Joules per nucleon) of 37Cl17 with a nuclear mass of 36.97031 amu
A:
Q: (7) Write a balanced nuclear equation for the following reactions and identify X, X, and y a) XCH-2,…
A:
Q: 1. Write balanced nuclear equations for a)the loss of an alpha particle by Th-230 b)the loss of a…
A: Alpha decay or α-decay is a type of radioactive decay in which the atomic nucleus emits an alpha…
Q: 5. Complete the following nuclear equations by filling in the correct symbols: 27 13AL + → Na + He…
A:
Q: Determine the identity of the daughter nuclide from the electron capture by CI - 36 Ar-37 K-38 O…
A:
Q: Write the nuclear equation represented by the transmutation reaction: 9Be (aplha,n) 12C
A: The given shorthand representation of a nuclear reaction states that an alpha particle is bombarded…
Q: 3. Complete the following nuclear equations and identify X in each case: 1. Mg + ip → ța + X 2. Co +…
A: a nuclear decay is type of radioactive decay where an unstable nuclide is converted to stable…
Q: 18. Samarium - 151 is a beta emitter. Write an equation for this nuclear reaction and the product…
A:
Q: Be sure to answer all parts. Complete the following nuclear equations and identify X in each case:…
A:
Q: Complete the following nuclear bombardment equation by filling in the nuclear symbol for the missing…
A:
Q: The uranium decay series from U-238 to stable lead (Pb-206) is: 238U → 234TH -→ 34P. 91 92 210Pb →…
A: α - particle emitters are- U92238 → T90234h + H24e U92234 → T90230h + H24e T90230h → R88226a + H24e…
Q: 1. Complete and balance these nuclear equations by supplying the missing particles: (а) 27 13А| +…
A: We can balance the radioactive reaction as below : We need to count the total mass number in…
Q: Write balanced nuclear equations for the following reactions and identify X: (a) X (p,a) 3c (b) AI…
A: a) X(p,α) 126C The balanced nuclear equation is, 157N + 11p ---> 126C + 42α So, X = 157N
Q: Write a balanced nuclear equation for the following reactions and identify X, X, and y a) XCH-2, H-)…
A:
Q: Potassium (Z = 19) has three primary isotopes: K-39, K-40, and K-41. The nuclear mass of K-40 is…
A:
Q: What is the balanced nuclear reaction for the following process? Iodine-131, used to treat…
A: It is given that iodine-131 is undergoing beta decay.
Q: Which of the following radioisotopes are most likely to be used in nuclear medicine? (write the…
A: Given information:For 40K half-life is 1.3 × 109 years.For 42K half-life is 12 hours.
Q: Nuclear scientists have synthesized approximately 1600 nuclei not known in nature. More might be…
A: Nuclear reaction is the one which takes place in the nucleus of an atom. Nuclear equation is…
Q: Complete the following nuclear fission reaction. *U+}n→3;n +3e + Te + 235 e 189 -1 92
A: Concept Atomic mass of reactant = Atomic mass of product Atomic no. Of reactant = Atomic no. Of…
Q: The nuclear mass of Ba141 is 140.883 amu. Calculate the binding energy per nucleon for Ba141
A: The mass number = 141 and the atomic number, Z = 56 ( no. of protons) The number of neutrons, N =…
Q: Write balanced nuclear equations for the following reactions and identify X: a. X (p,a) 1C d. 9Se…
A: Hey, since there are multiple subparts posted, we will answer first three subparts. If you want any…
Q: the equations for the following induced nuclear reactions: (a) As (d, 2n) X? (b) iMg (d, a) X? (c)…
A:
Q: Calculate the nuclear binding energy (J) and the nuclear binding energy per nucleon of (208.9804…
A:
Q: 35 16 S, 8. 214 Bi 9. 83 + Y 27 10. Si 14 +1 ↑
A:
Q: Complete the following nuclear equation by identifying X. (Enter the complete nuclear equation.) 59…
A: C2759o +H12→C2760o+ X Total number of protons in reactants = 27+1 = 28 Total number of protons in…
Q: Which of the following correctly balances this nuclear fission reaction? 1n + 235U = 131I _ +3 0 n…
A:
Q: Identify the missing species in the nuclear equation given 2655FE+ -10E -> ? 2753FE 2555Mn…
A: 2655Fe is the radio-active isotope of iron. The nucleus of this radioactive isotope contains 29…
Q: 8. He + 27AI ---→ H + 2 13
A: In balanced nuclear equation, the sum of the mass numbers and the sum of the atomic numbers balance…
Q: Complete each of the following nuclear decay reactions. -je 4C → B + 225 89Ac - 221 87 Fr + 13M9 →…
A: A nuclear reaction involves release of energy either by fission or by fusion.
Q: Q2 The most stable nucleus (in terms of binding energy per nucleon) is 56Fe. Based on this, what…
A:
Q: 10.1 Palladium-107 (Z = 46) undergoes beta (B) decay (t1/2 = 6.5 x 105 yr). Write the balanced…
A:
Q: Be sure to answer all parts. 237 Np decays by a-particle emission and has a half-life of 2.20 × 10°…
A: We have given 23793Np decays by a-particle emission and has a half-life of 2.20 × 106 years. (a)…
Q: it can 5. Complete the following nuclear equations by filling in the correct symbols: 27 134 Al + Na…
A: When the unstable nuclei of the metals release energy/radiations i.e. are alpha, beta and gamma rays…
Q: 19. Complete the following nuclear equations:| 27A1 13 He + 241 'Am 95 + 241 Pu 94 + 6 'n 238U 12C +…
A: The reactions are given known as nuclear reaction, because new nuclide forms from a old nuclide.…
Q: Complete the following nuclear equations. 224, *Fr + 87 210 Bi → 83 1° 231 Ра 91 227 Ас + 89 238U →…
A: In nuclear reaction radioactive element undergoes radioactive decay and produce non radioactive…
Q: Fill in the missing particle in each nuclear equation. At + He 217 a. b. 24Pu Na d. se + 241 5Am +…
A: For this question we will use the concept of mass no and atomic no. we know that, i)the sum of…
Q: Rn - 220 as a nuclear equation
A: 220Rn undergoes nuclear reaction and it emits one α particle to along with gives 216Po which is…
Q: 50Sn126 is an example of OA) fusion OB) fission OC) gamma emission OD) beta decay 51Sb¹26 +?
A: Nuclear fission is the process in which when the nucleus of a heavy atom is bombarded with neutrons,…
Q: Complete the following nuclear bombardment equation by filling in the nuclear symbol for the missing…
A: Nuclear reaction: The given reaction is a nuclear reaction. In the nuclear reaction, one nuclide is…
Q: Complete the following two balanced nuclear equations. Hint: You need to check the periodic table.…
A: Given, The complete balanced nuclear equations are:
Q: Nuclear transmutation - 239Pu(α, n), what is the product? a. Cm-245 b. Cm-242 c. U-242 d. U-243…
A: Nuclear reaction: A nuclear reaction must cause a transformation of at least one nuclide to another.…
Q: 2. Complete the following nuclear equation and identify X in each case: 4 a + X 2" 26 a.) Mg 12M9 +…
A: Since you have asked multiparts questions, we will solve the first three sub parts questions for…
Q: The following reaction represents what nuclear process? 131 53 0 -1 e + 131 54 Xe neutron…
A: We have to predict the nuclear process.
Q: 2. Complete the following nuclear equation and identify X in each case: a.) Ma + ip 26, а.) 12 4 2a…
A:
Q: 4. Complete the following nuclear equations, and identify X in each case. a. 222Rn → 218Po + X b.…
A: We are authorized to answer three subparts at a time, since you have not mentioned which part you…
Q: Complete the following nuclear equations and identify X in each case: 135 135 (a) Xe +X 53 54 X = 40…
A: In the nuclear reaction, the total atomic number and total mass number should be equal.
Q: Complete the following nuclear bombardment equation by filling in the nuclear symbol for the missing…
A: In a balanced nuclear equation sum of atomic number and sum of mass number is same on both side of…
Q: The uranium decay series from U-238 to stable lead (Pb-206) IS: 238U → 234Th → “Pa → 92 U - Th →…
A: alpha- particle emission is accompanied by decrease in mass no. by 4 and decrease in atomic no. by…
Q: a Choose the correct balanced nuclear equations for the beta decay of each of the following…
A: Since you posted multiple question we answered the first one. We know that beta decay is a type of…
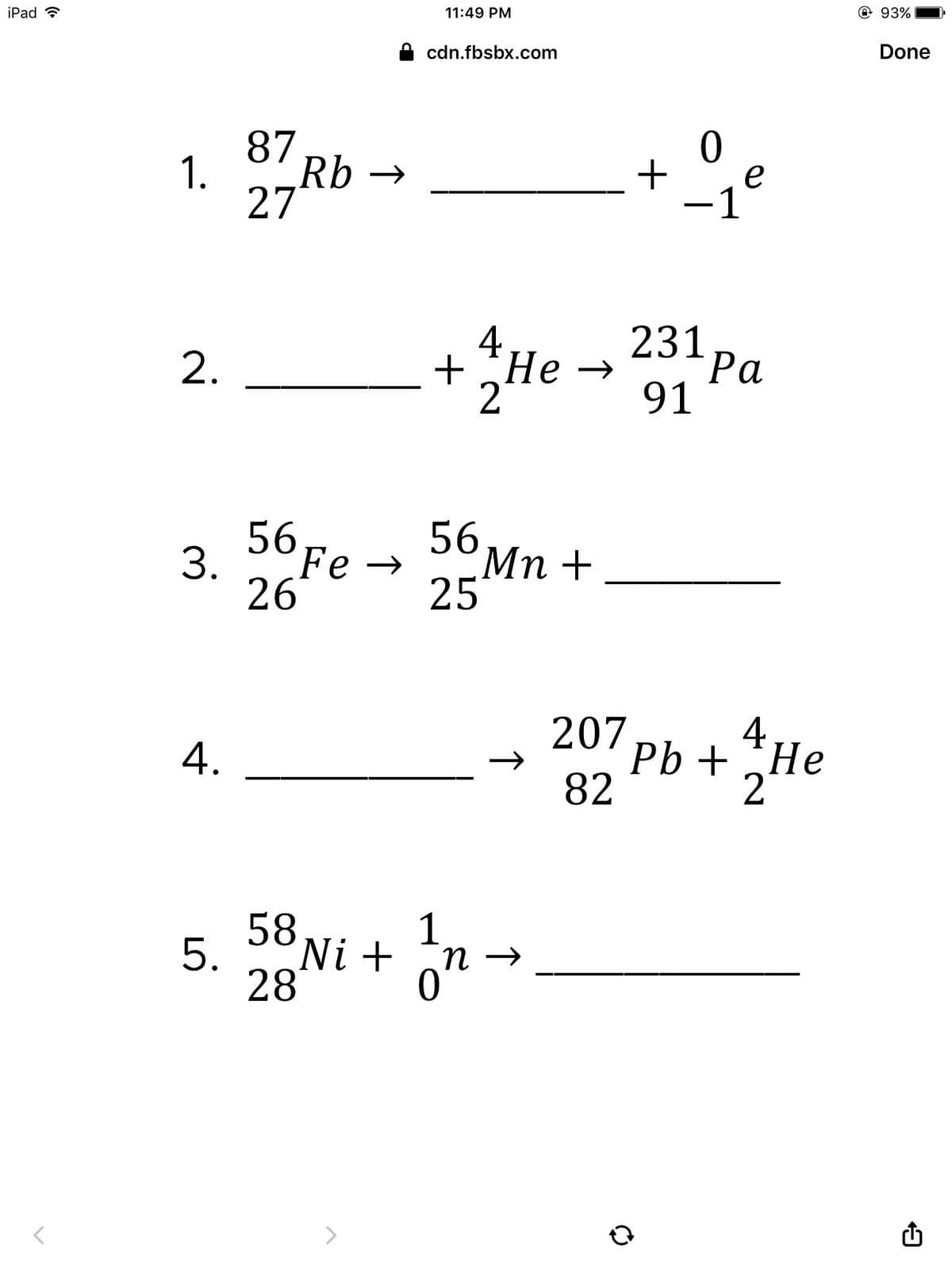
Direction: Complete the following
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

- Give advantages of Ziegler–Natta catalysts ?The maximum safe [Ba2+]=1.5x10-5M. Maintaining 1.0x10-3M levels for each of the anions below would ensure that this level is achieved except (Ksp of BaF2=1.7x10-6, BaCO3=5.9x10-9, BaC2O4=7.4x10-14, and BaSO4=1.1x10-10) A SO42- B C2O42+ C CO32- D F-Ali has measured the oxidative stability of palm oil sample under accelerated storage condition at 60*C for 6 days The following data were recorded for the peroxide value and p anisidine value of the palm oil after 5 days of storage time: Sample weight 1.0030 g Concentration of NazSzO3 = 0.001 N Volume of NazSzO3 titrated for sample solution = 1.35 ml Volume of NazSzO3 titrated for blank solution 0.20 ml Absorbance for sample solution = 0.907 Absorbance for blank solution 0.203 Calculate: i) peroxide value ii) p-anisidine value iii) TOTOX value
- Elaborat ur answer pls thank uWhat transition (d–d charge transfer) is responsible for the dark blue color of [Cu(phen)2]BF4? Why does the dissolution [Cu(phen)2]BF4 of in dichloromethane (DCM) result in a dark orange solution? Justify your answer.Three large proteins are ionized at the pH at which an electrical FFF separation is carried out. (i) if the ions are designated A2+, B2+, and C3+, predict the order of elution. (ii) What is FFF? (iii) List the four subtechniques of FFF.
- The Racah parameter B for [Co(CN)6]3−=460cm−1, and [Co(NH3)6]3+= 615cm−1. Explain the difference in nephelauxetic effect. Thank you!Generally high temperature is favourable for chemisorption.’ Why?The formation constant for the complexation of Mg2+ by EDTA is logKflogKf = 8.79. The conditional formation constant (Kf’) at pH 8 (αY4−αY4−= 4.2 x 10-3) is: 0.037 2.6 x 106 5.14 1.5 x 1011 6.4
- Racah parameter B for [Co(CN)6]3−=460cm−1, and [Co(NH3)6]3+= 615cm−1. Explain the difference in nephelauxetic effect.The following data were obtained on the initial rates of reaction of a d-metal complex in aqueous solution. For the experiments a. [Y] = 2.7 mmol/dm3, and for experiments b. [Y] = 6.1 mmol/dm3 Conc. complex/(mmol dm-3) 8.01 9.22 12.11 Exp. a. v/ (mol dm-3 s-1) 125 144 190 Exp. b. v/ (mol dm-3 s-1) 640 730 960 b) use the data above to calculate the rate constant. k = ________ (mol/L/s, s-1, L/mol/s, or L2/mol2/s). 3 s.f. normal format.A chemist obtained the following data for percent lindane in the triplicate analysis of an insecticide preparation: 7.23, 6.95, and 7.53%. Calculate the 90% confidence interval for the mean of the the three data, assuming that (a) the only information about the precision of the method is the precision for the three data. (b) on the basis of long experience with the method, it is believed that s---->σ lindane. (c) If s=0.28 is good estimate of σ, how many replicate measurement should be made in order for the mean for the analysis of sample to be within 0.2% of the true mean 90% of the time.

