Q: Provide the necessary compounds to form heptane according to the given Ether A 2 Li B Pentane Cul -…
A: Given: We have to make heptane for the given reaction.
Q: Give the mechanism with orrect agents 9 0 Br
A:
Q: ructural formula of an alkene that undergoes acid-catalyzed hydration to give 2-butanol as a major…
A: The corresponding reactant and mechanistic aspects of the given reaction are discussed below. Acid…
Q: 11. (challenge problem) A 6,355 gram ingot of uranium (c=0.12 J/g-°C) has a temperature of 1050°F…
A: Given: Mass of ingot of uranium = 6355 g Initial temperature of uranium = 1050°F Heat capacity of…
Q: 1. At high temperatures, sulfur combines with iron to form the brown-black iron (II) sulfide: Fe(s)…
A: Substance which is present in low amount consumes firstly during reaction are called as limiting…
Q: Be sure to answer all parts. Complete the reactions to show the synthesis of the following compound…
A: Detail mechanistic pathway is given below to carry out the following conversion
Q: Why do we have to measure the boiling point of distilled water during the colligative properties…
A: Colligative Properties depends Upon the Number of solute particles and independent of identity of…
Q: 3. Verify that the wave function y(x) = Ae (ku) ¹/² x² /(2n)] (where A, m, and k are all constants)…
A: The Schrodinger statement is given by H^ψ(x) = Eψ(x) Where, H^ is the Hamiltonian operator…
Q: Click the "draw structure" button to launch the drawing utility. Draw the two enantiomers produced…
A:
Q: How many isomers, both structural and geometric, have the molecular formula C5H12?
A:
Q: Which of the following reactions is a substitution reaction? C6H6 + Cl2 → C6H5Cl + HCl CH2=CH2 + Cl2…
A:
Q: a. Write the bondline structure of the osazone produced by glucose. b. Identify two monosaccharides…
A:
Q: Determine the equilibrium constant (Keq) at 25°C for the reaction Cl₂(g) + 2Br¯ (aq) = 2Cl¯ (aq) +…
A:
Q: (c) To study the kinetics of iodination of acetone, why starch is used as an indicator just before…
A: Introduction : Iodometric titration is the type of titration in which an oxidizing agent is…
Q: Fill in the blanks: Br H₂C. CH₂ CH₂ The major product of the elimination of HBr from the above…
A: Given: Elimination reaction of .
Q: What volume in liters does 0.137 g of O₂ occupy at 27.2 °C and 755 torr? Assume the standard…
A:
Q: LOH Na "OH Which will best explain why the reaction is not likely to happen? Select the correct…
A: Nature of fluorine as leaving group in Substitution reaction
Q: 4) For F₂ molecule: a) b) d) e) Construct a MO diagram. State the HOMO and LUMO. Calculate the bond…
A: Atomic orbitals of two fluorine atoms combine to form molecular orbitals.
Q: 1. Consider a situation where two volumes of water, one hot, one cold, are both added to a foam cup.…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: Calculate the AH for the reaction NO(g) + O(g) given the following information: NO₂(g) NO(g) + O3(g)…
A: Given aim equation is : Step equations are : Calculate the change in enthalpy of the aim equation…
Q: What is the predicted freezing point of water? * Why do you think you need to put salt in the…
A: The need for an ice bath is to keep the temperature of the system as low as the freezing point of…
Q: Be sure to answer all parts. A³H nucleus decays with an energy of 0.01856 MeV. Convert this energy…
A: Here we are required to find convert the energy for 3H decay from mega electron volume to electron…
Q: Pls help ASAP.
A: A question based on environmental pollution that is to be accomplished.
Q: (f) Why alternating current is used for conductometric measurements? (g) What will happen if large…
A: h) the integrated rate equation for a first order reaction is given by, ln[A] = -kt+ln[A0] Where [A]…
Q: Provide the structure of the major organic product(s) in the following reaction. CH3 Cl₂ AICI 3
A: Since you have posted multiple questions as per guidelines we can answer only one per session. If…
Q: To study the kinetics of iodination of acetone, why starch is used as an indicator just before the…
A: Introduction : Iodometric titration is a volumetric chemical analysis.Iodometric titration is…
Q: For the following reaction, use the plots below to determine the reaction order wit respect to NO2…
A: Given : We have to tell the order of NO2 .
Q: A solute was added to a solvent and mixed well. Some of the solute settled to the bottom of the…
A: A solute was added to a solvent and mixed well. Some of the solute settled to the bottom of the…
Q: Give the mechanism and choose correct option H30t heat ? OCH 3 10 A B C 애 김선 E
A: We have to predict the product of given reaction.
Q: Which one of the following will be most acidic and why? 2-Chlorobutanoic acid, 4-Chlorobutanoic…
A: 2-Chlorobutanoic acid is most acidic in nature.
Q: Which of the following alcohols is least likely to be soluble in water? 3-pentanol 2-butanol…
A:
Q: TAMILTUNE Copyright © 1994 M 24 Proton Nha 200 190 Carbon 13 NMR N WIRE VIVEKPRIERS 24 -1007.05 MUD…
A: C11H14O (DBE = 11 - (14/2)+1 = 12-7 = 5 = 4+1. One benzene ring and one carbonyl. Therefore the…
Q: Anilinium chloride (C6H5NH3Cl) is a weak acid (Ka = 2.4 x 10-5). A sample of anilinium chloride…
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: ENTROPY, MICROSTATES, AND PROBABILITY Overview Expansion of a gas at the molecular level is…
A:
Q: A 21.2 ml sample of 0.283 M trimethylamine, (CH3)3N, is titrated with 0.328 M hydrobromic acid. At…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Maple syrup is made from the sap of a maple tree. Whe the sap is tapped from the tree it is 4.0% by…
A: Here we are required to find the mass of sap that would be needed for making 1kg of syrup
Q: 1.Common Ion Effect: The solubility of a material is unchanged when at least one of its conjugates…
A: We are authorised to solve only first three subparts of a question. Please post rest of the subparts…
Q: Using only ethane and other 2 carbon reagents as the starting material propose a synthesis of OH…
A: Detail synthetic route is given below
Q: What is the point group for NH3?
A:
Q: Solution where more solute can still be dissolved at the given temperature. Supersaturated…
A: Here in we discuss about the behaviour of the solution upon increasing the amount of solute getting…
Q: Draw a Haworth projection of a common cyclic form of this monosaccharide: 0 НО H H OH HỌ-H НО- h…
A:
Q: Consider these two reactions: Reaction 1: 20F₂(g) → O₂(g) + 2 F₂ (g) AH = -49.4 kJ Reaction 2: N₂(g)…
A: ★ Answer : Reaction 2 is non spontaneous. ★ Explanation :
Q: A. Predict whether AS is positive or negative by considering disorder in the reactants and products.…
A: ∆S is positive when entropy of product is more than entropy of reactant. ∆S is negative when…
Q: Fe(s) + H₂O(g) CO2(g) + H₂O(1) System FeO(s) + H₂(g) H₂CO3(aq) at 20°C с P F Intensive Variables
A: Answer: This question is based on phase rule which is shown below: F=C-P+2 Here: F=degree of…
Q: Identify the following as pure substance, homogeneous solution, heterogeneous mixture or colloidal…
A: We have to identify which one is pure substance, homogeneous solution, heterogeneous mixture or…
Q: Choose the correct reactants for the following multistep reaction: ? O :O CI
A:
Q: For the cell shown, the measured cell potential, Ecell, is -0.3619 V at 25 °C. Pt(s) | H₂(g, 0.721…
A: Given -> Ecell = -0.3619V Pressure of H2 = 0.721 atm Concentration of Cd = 1.00 M
Q: how do i know if my measurement is significantly different from the exact value, at least 2…
A: Given: Exact value : 0.25 Average = 0.223 Standard deviation = 0.025 RSD = 11.2 %
Q: The complex trans-[Co(en)2Cl2]+ undergoes substitution of Cl- by H2O slower than the complex…
A: To explain: The complex trans-[Co(en)2Cl2]+ undergoes substitution of Cl- by H2O slower than the…
Q: The geometry favors the E2 mechanism because it will promote the effective
A: The leaving group is always anti-periplanar to the hydrogen that is removed on the β carbon. in…
Identify the product in the following reactions
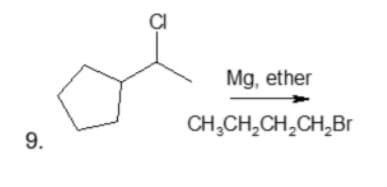
Step by step
Solved in 2 steps with 2 images


