9. Nitrogen monoxide is formed in automobile exhaust by the reaction of N2 and O2 from air, and produced by the following reaction: N2(g) + O2(g) <==> 2NO(g). The equilibrium constant for this reaction is 1.23 x 10-2 at 135°C. What is the direction of the reaction when a mixture that is 0.80 M N2, 0.60 M O2, and 0.10 M NO gets near equilibrium at 135°C?
9. Nitrogen monoxide is formed in automobile exhaust by the reaction of N2 and O2 from air, and produced by the following reaction: N2(g) + O2(g) <==> 2NO(g). The equilibrium constant for this reaction is 1.23 x 10-2 at 135°C. What is the direction of the reaction when a mixture that is 0.80 M N2, 0.60 M O2, and 0.10 M NO gets near equilibrium at 135°C?
General, Organic, and Biological Chemistry
7th Edition
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:H. Stephen Stoker
Chapter9: Chemical Reactions
Section: Chapter Questions
Problem 9.76EP: Calculate the value of the equilibrium constant for the reaction N2(g)+2O2(g)2NO2(g) if the...
Related questions
Question
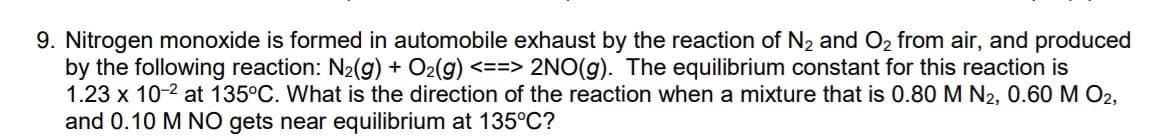
Transcribed Image Text:9. Nitrogen monoxide is formed in automobile exhaust by the reaction of N2 and O2 from air, and produced
by the following reaction: N2(g) + O2(g) <==> 2NO(g). The equilibrium constant for this reaction is
1.23 x 10-2 at 135°C. What is the direction of the reaction when a mixture that is 0.80 M N2, 0.60 M O2,
and 0.10 M NO gets near equilibrium at 135°C?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning