9. What is the mass of one-half mole of SO2? а. 8 g с. 24 g b. 12 g d. 32 g 10. How many grams are there in 1 mole of Zinc? а. 30 g c. 65.38 g b. 60. 2 g d. 130.76 g
9. What is the mass of one-half mole of SO2? а. 8 g с. 24 g b. 12 g d. 32 g 10. How many grams are there in 1 mole of Zinc? а. 30 g c. 65.38 g b. 60. 2 g d. 130.76 g
Chapter4: Calculations Used In Analytical Chemistry
Section: Chapter Questions
Problem 4.14QAP
Related questions
Question
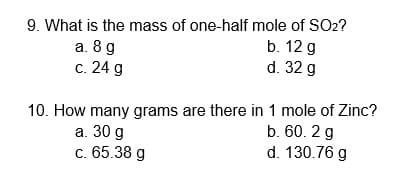
Transcribed Image Text:9. What is the mass of one-half mole of SO2?
b. 12 g
a. 8 g
с. 24 g
d. 32 g
10. How many grams are there in 1 mole of Zinc?
а. 30 g
c. 65.38 g
b. 60. 2 g
d. 130.76 g
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning
