9. Which element is oxidized in the following reaction: 2FECI2 + Cl2 → 2FECI3 if Fe goes from +2 to +3 and CI goes from 0 to -2? Fe Neither element CI Cl and Fe
9. Which element is oxidized in the following reaction: 2FECI2 + Cl2 → 2FECI3 if Fe goes from +2 to +3 and CI goes from 0 to -2? Fe Neither element CI Cl and Fe
Chemistry for Engineering Students
3rd Edition
ISBN:9781285199023
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter1: Introduction To Chemistry
Section: Chapter Questions
Problem 1.6PAE: Use the web to determine the differences in the amounts of aluminum recycled in states where there...
Related questions
Question
100%
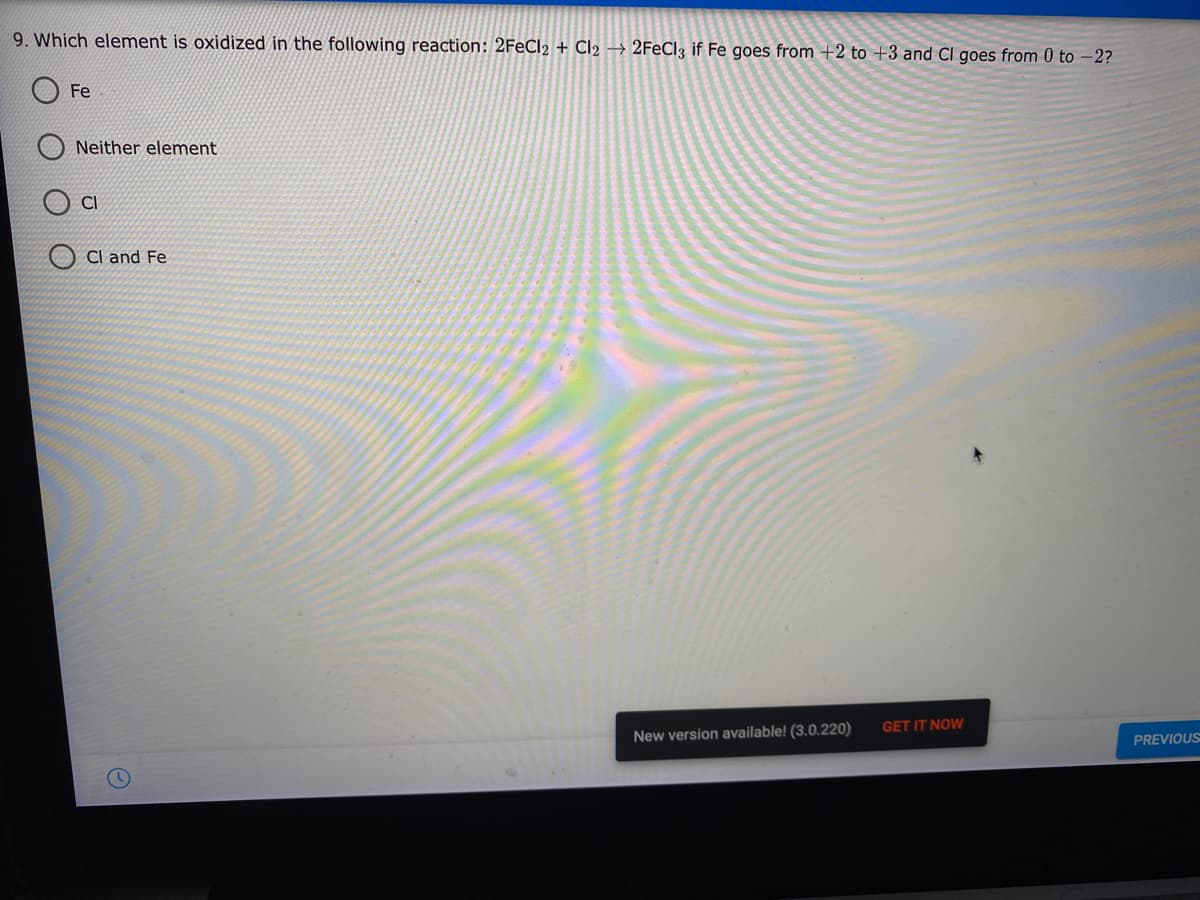
Transcribed Image Text:9. Which element is oxidized in the following reaction: 2FECI2 + Cl2 → 2FECI3 if Fe goes from +2 to +3 and CI goes from 0 to –2?
Fe
Neither element
CI
Cl and Fe
GET IT NOW
New version available! (3.0.220)
PREVIOUS
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning