9. Which structure represents a portion of the repeating unit for the polymer formed from cationic polymerization of methyl vinyl ether? (A) BC13 H₂O (B) (C) (D) faborat галаха n
9. Which structure represents a portion of the repeating unit for the polymer formed from cationic polymerization of methyl vinyl ether? (A) BC13 H₂O (B) (C) (D) faborat галаха n
Chapter31: Synthetic Polymers
Section31.SE: Something Extra
Problem 21AP
Related questions
Question
Please explain the answer, and provide a mechanism if possible
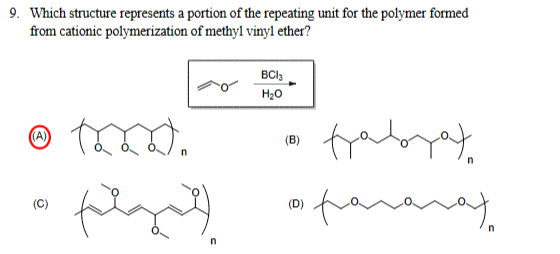
Transcribed Image Text:9. Which structure represents a portion of the repeating unit for the polymer formed
from cationic polymerization of methyl vinyl ether?
(A)
BC13
H₂O
(B)
(C)
(D)
faborat
галаха
n
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 1 steps with 1 images

Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning