A 0.0303-m container is initially evacuated. Then, 5.71 g of water is placed in the container, and, after some time, all of the water evaporates. If the temperature of the water vapor is 448 K, what is its pressure? Number Units
A 0.0303-m container is initially evacuated. Then, 5.71 g of water is placed in the container, and, after some time, all of the water evaporates. If the temperature of the water vapor is 448 K, what is its pressure? Number Units
College Physics
1st Edition
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:Paul Peter Urone, Roger Hinrichs
Chapter13: Temperature, Kinetic Theory, And The Gas Laws
Section: Chapter Questions
Problem 59PE: Air in human lungs has a temperature of 37.0C and a saturation vapor density of 44.0g/m3. (a) If...
Related questions
Question
Transcribed Image Text:Current Attempt in Progress
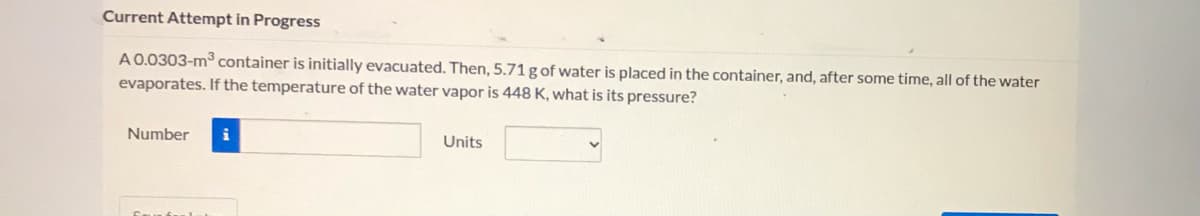
A0.0303-m container is initially evacuated. Then, 5.71 g of water is placed in the container, and, after some time, all of the water
evaporates. If the temperature of the water vapor is 448 K, what is its pressure?
Number
i
Units
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning


College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning



Glencoe Physics: Principles and Problems, Student…
Physics
ISBN:
9780078807213
Author:
Paul W. Zitzewitz
Publisher:
Glencoe/McGraw-Hill

College Physics
Physics
ISBN:
9781305952300
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning