A 5.71 g sample of graphite was heated with 67.7 g of O₂ in a 8.00 L flask. The reaction that took place was C (graphite) + O₂(g) → CO₂(g) After the reaction was complete, the temperature in the flask was 182 °C. What was the total pressure inside the flask Be sure your answer has the correct number of significant digits. atm x10 X
A 5.71 g sample of graphite was heated with 67.7 g of O₂ in a 8.00 L flask. The reaction that took place was C (graphite) + O₂(g) → CO₂(g) After the reaction was complete, the temperature in the flask was 182 °C. What was the total pressure inside the flask Be sure your answer has the correct number of significant digits. atm x10 X
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter6: The Gaseous State
Section: Chapter Questions
Problem 6.7QE
Related questions
Question
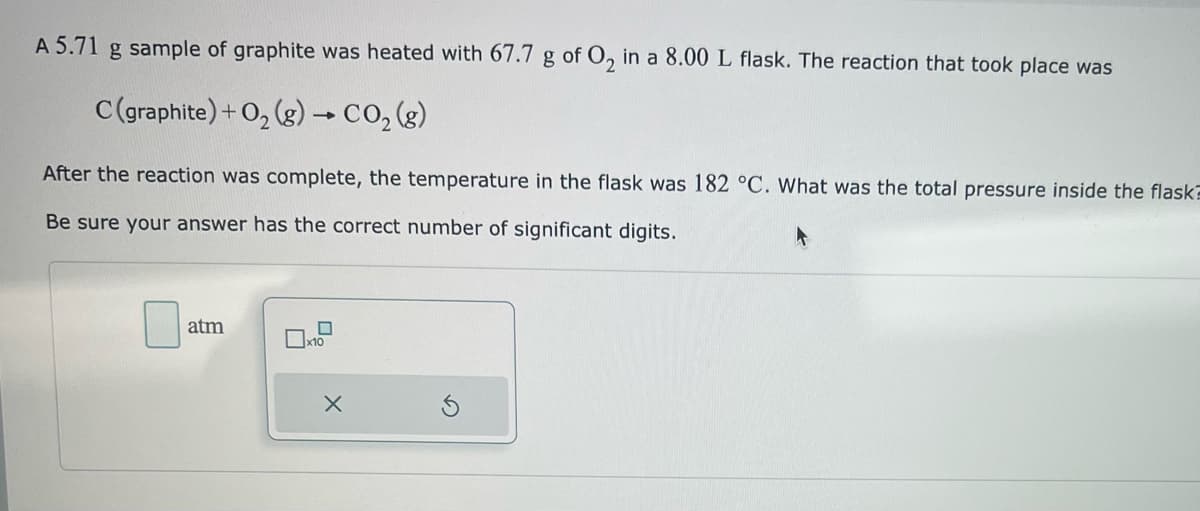
Transcribed Image Text:A 5.71 g sample of graphite was heated with 67.7 g of O₂ in a 8.00 L flask. The reaction that took place was
C (graphite) + O₂(g) → CO₂(g)
After the reaction was complete, the temperature in the flask was 182 °C. What was the total pressure inside the flask
Be sure your answer has the correct number of significant digits.
atm
x10
X
Expert Solution
Step 1
Given:
Mass of graphite = 5.71 g
Mass of O2 = 67.7 g
Volume = 8.00 L
Temperature in the flask = 182 °C
To find:
Total pressure,P = ?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning