A 52.0-mL volume of 0.35 M CH3 COOH (Ka = 1.8 x 10-5) is titrated with 0.40 M NaOH. Calculate the pH after the addition of 25.0 mL of NaOH. Express your answer numerically. • View Available Hint(s) ? pH =
A 52.0-mL volume of 0.35 M CH3 COOH (Ka = 1.8 x 10-5) is titrated with 0.40 M NaOH. Calculate the pH after the addition of 25.0 mL of NaOH. Express your answer numerically. • View Available Hint(s) ? pH =
Chapter7: Neutralization Titrations And Graphical Representations
Section: Chapter Questions
Problem 7P
Related questions
Question
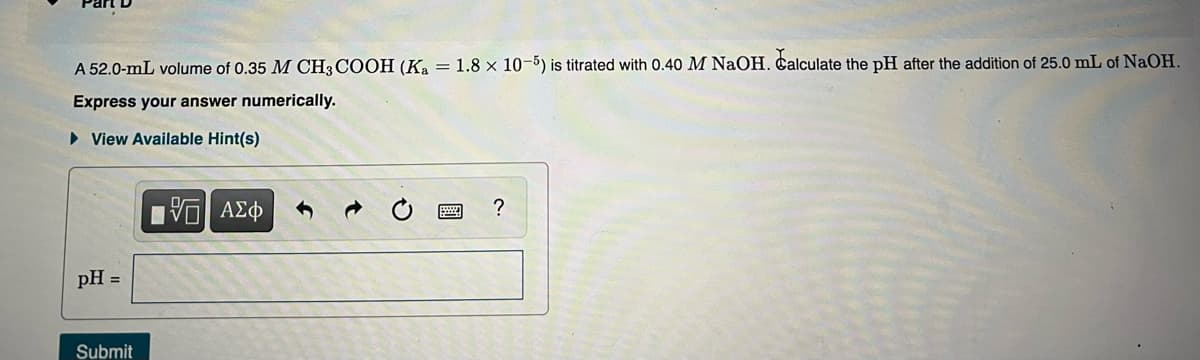
Transcribed Image Text:A 52.0-mL volume of 0.35 M CH3 COOH (Ka = 1.8 × 10-5) is titrated with 0.40 M NaOH. Calculate the pH after the addition of 25.0 mL of NaOH.
Express your answer numerically.
• View Available Hint(s)
?
pH =
Submit
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you



Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning



Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning