A 800 mL sample of nitrogen (N2) was col- lected by displacement of water at 24°C un- der a total barometric pressure of 1075 torr. What mass of dry nitrogen was collected? The vapor pressure of water at 24°C is 22 torr. Answer in units of g.
A 800 mL sample of nitrogen (N2) was col- lected by displacement of water at 24°C un- der a total barometric pressure of 1075 torr. What mass of dry nitrogen was collected? The vapor pressure of water at 24°C is 22 torr. Answer in units of g.
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Chapter5: The Gaseous State
Section: Chapter Questions
Problem 5.145QP: A 275-mL sample of CO gas is collected over water at 31C and 755 mmHg. If the temperature of the gas...
Related questions
Question
Please answer both questiosn as we are allowed to ask 3 questions everytime

Transcribed Image Text:A 800 mL sample of nitrogen (N2) was col-
lected by displacement of water at 24°C un-
der a total barometric pressure of 1075 torr.
What mass of dry nitrogen was collected? The
Your
vapor pressure of water at 24°C is 22 torr.
Answer in umits of g.
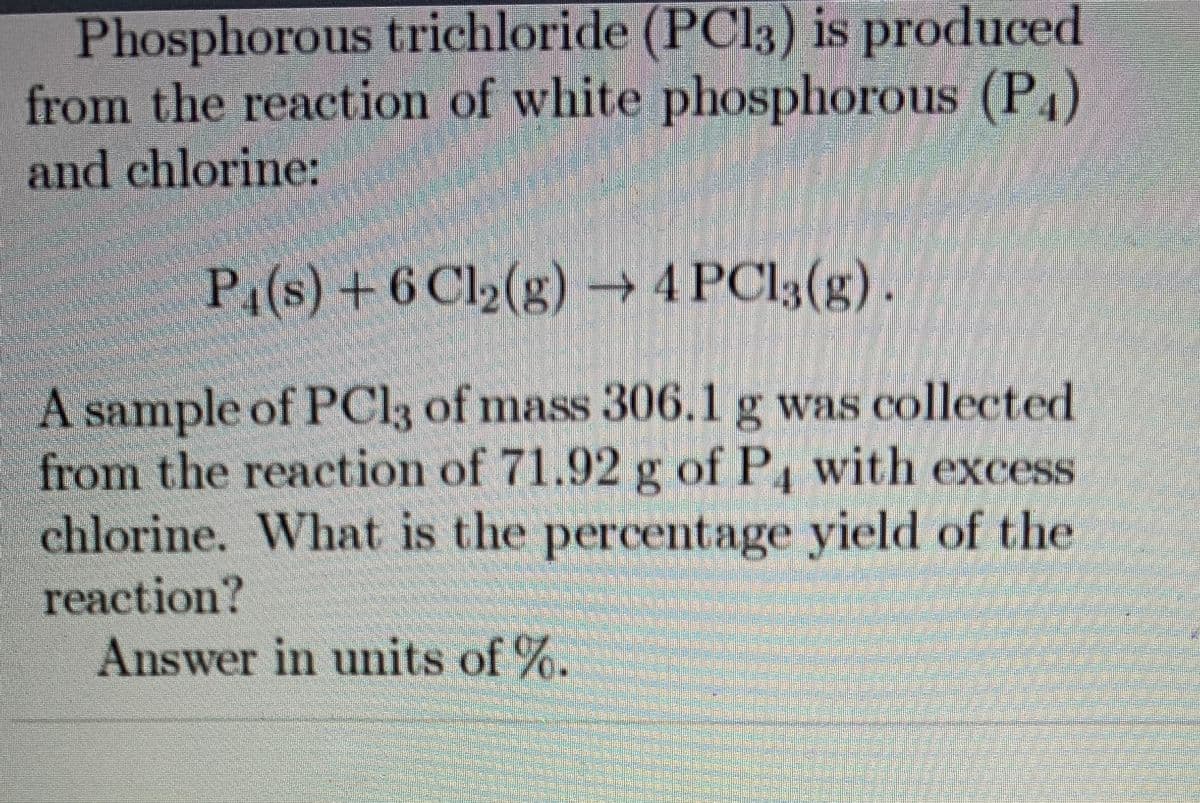
Transcribed Image Text:Phosphorous trichloride (PC13) is produced
from the reaction of white phosphorous (P4)
and chlorine:
P1(s) +6 Cl2(g) → 4 PCI3(g).
|A sample of PCI3 of mass 306.1 g was collected
from the reaction of 71.92 g of P4 with excess
chlorine. What is the percentage yield of the
reaction?
Answer in units of %.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax
