Q: 1. Draw the Product (If applicable) of the Following Reactions: HCI + CI Br
A: The first reaction has a carbonyl compound. A carbonyl carbon center is electron-deficient and acts…
Q: IV. A student isolated chemical A and B using column chromatography based on the TLC result sh…
A: The objective of the question is to calculate the Retention Factor and answer the questions related…
Q: HOOC H H CH2NH2 Tranexamic acid, a drug useful against blood clotting, is prepared commercially from…
A:
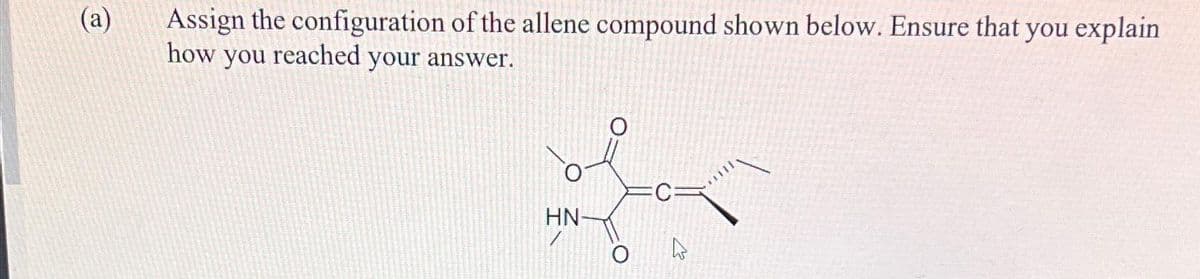
Q: 20
A: The objective of the question is to devise a reaction scheme to form (S)-2-deuterobutanenitrile from…
Q: Check the box under each molecule in the table below that is an isomer of this molecule: If there…
A: Given,The molecules:
Q: Label all of the nucleophilic and electrophilic sites on the following molecule
A: A question based on introduction to organic chemistry.. For the given molecule, the nucleophilic and…
Q: Classify each element below as belonging to the representative elements, transition elements, or…
A: The periodic table is organized into three main categories: representative elements, transition…
Q: FO/F The Stern Volmer plot shown below is a plot of relative fluorescence intensity (Fo/F) of…
A: The Stern-Volmer equation is a mathematical relationship used to describe the quenching of…
Q: Please explain in detail whats going on in this slide Strong vs Weak Acids A strong acid is almost…
A:
Q: Draw the simplest mechanism possible for the reaction below. You may need to re-draw structures to…
A: Possible Mechanism: Need to be drawn
Q: OH Provide a reasonable synthesis of 1-cyclohexyl-1-ethanol starting from cyclohexanol. OH
A: The objective of this question is to provide a reasonable synthesis of 1-cyclohexyl-1-ethanol…
Q: If a buffer solution is 0.110 M in a weak acid (K₁ = 6.6 x 10-5) and 0.420 M in its conjugate base,…
A: To find the pH of a buffer solution, you can use the Henderson-Hasselbalch equation, which is given…
Q: Draw mechanism using Curved arrows to show how each product is formed. Label each stop as (1) Proton…
A:
Q: Please show the second step of the mechanism properly.
A: p-Toluenesulfonic acid also called tosylic acid (TsOH) is an organic acid. It is a strong acid…
Q: How many milliliters are required of 0.7M HCI to make a titer of 0. IM solution?
A: The objective of the question is to determine the volume of needed.It is given that,The molarity of…
Q: Complete the following reaction scheme. Pay attention to stereochemistry. Part 1 of 3 OH H OTSOH,…
A: The -OH group of alcohol is a poor leaving group, so must be converted to good leaving group.
Q: H2SO4 (aq) + E → H2O(1) + O2(g) + SO2(g)
A: A chemical reaction where the count of atoms participating in the reaction is same on both sides of…
Q: Benzoic acid is monoprotic with a K₁ of 6.5x10-5. What is the pH of a 0.00287 M aqueous solution of…
A: The objective of this question is to calculate the pH of a 0.00287 M aqueous solution of benzoic…
Q: Please explain your work 1. There is a non-electrically excitable cell in a bath at room temperature…
A: conducatnces of Na+=I/V =>not possibleconductances of k+= 60/-60 =>-1conductances of cl-…
Q: Determine AG (in kJ) for the following reaction at 325 K when the concentrations of the species are…
A: The objective of this question is to calculate the Gibbs free energy change (ΔG) for the reaction of…
Q: How many significant figures are in 0.005239800 mL? 8 9 O 10 5 7
A: The question is asking for the possible subshells for the principal quantum number n = 4 in an atom.…
Q: Rank the nucleophiles in the order of increasing strength. Choose 1 for the weakest nucleophile and…
A: Electron rich species are acts as nucleophiles. Nucleophiles are Lewis bases. Nucleophiles attack on…
Q: Deterioration of buildings, bridges, and other structures through the rusting of iron costs millions…
A: The balanced reaction taking place is given as,
Q: In an aqueous solution, which pair of compounds in equimolar concentrations best represents a pH…
A: The objective of the question is to identify which pair of compounds in equimolar concentrations…
Q: What is the freezing point, in \deg C, of a solution made with 1.15 mol of CHCI, in 530.0 g of CCI,…
A: Given data , Moles of CHCl3 = 1.15 moles for CHCl3 van't Hoff factor (i) = 1Mass of solvent (CCl4) =…
Q: What is the source of the nucleophilic attack? How does the oxygen get protonated? What causes the…
A: Here nucleophilic attack , protonation and elimination occur in the given reaction.
Q: Methanol, CH, OH, formerly known as wood alcohol, is manufactured commercially by the following…
A: moles of CO = 0.1470 molmoles of H2 = 0.3900 molmoles of CO at equilibrium = 0.1340 mol
Q: Consider the reaction CO(g) +Cl₂(g) →COCI₂(g) Use the standard thermodynamic data in the tables…
A:
Q: Draw out the equilibrium reaction for the 'flipping' of the cis-1,4-dibromocyclohexane (draw a chair…
A: Due to the high-angle strain, the planar structure of cyclohexanes is highly unstable and hence they…
Q: hos H H₂O
A: The given reactant is conjugated diene. A double bond acts as a nucleophile and extracts the acidic…
Q: A student reported the following composition of an unknown mixture: 25% NH4Cl, 30% NaCl, and 50%…
A: The objective of this question is to show the unknown mixture reported by the student exceed ,…
Q: Problem 1 (12pt) In an ideal Otto cycle of air, suppose the air is compressed to 650 K and 1800 kPa…
A: The objective of the question is to understand the processes involved in an ideal Otto cycle, sketch…
Q: Assuming an integrated luminosity of 100 fb is collected at the ATLAS detector at the LHC, calculate…
A: The aim of this question is to explain the integrated luminosity of 100 fb(-1) of the ATLAS…
Q: For each pair of substrates below, choose the one that will react faster in a substitution reaction,…
A: It is given that the products are a roughly 50/50 mixture of enantiomers. So, it is SN1 reaction.…
Q: Answer all. Explain briefly how these nuclear synthetic pathways aid in the production of heavy…
A: The aim of this question is to explain the nuclear fusion pathways contribute to the production of…
Q: please draw detailed mechanism. mention which one is the nucleophile, electrophile,base/ acid. Also…
A: Given is a Horner Wadsworth Emmond Wittig reaction.To find the mechanism of the reaction.
Q: Br EM 100 3.1.0 1.NaCN 2 HO SOCI, pyridine
A: Grignard reagent (R-MgX) is an organometallic reagent and is a source for carbanion as well as…
Q: An excited state hydrogen atom whose electron is 2p will be easier to ionize than an excited state…
A: The objective of the question is to understand why a 2p electron in an excited state hydrogen atom…
Q: Mass of pure aspirin expected: Mass of crude aspirin obtained: Percent yield in crude product = 2.72…
A:
Q: In any balanced chemical equation, the number of each type of atorn on both sides of the equation is…
A: The objective of this question is to balanced chemical equation represents a chemical reaction in…
Q: In the determination of ethanol concentration in alcoholic beverages using the GCFID method, why do…
A: The objective of this question is to show the determination of ethanol content is essential for…
Q: A voltaic cell is constructed in which the following cell reaction occurs. The half-cell…
A: Given, The cell reaction:Sn2+(aq) + Cd(s) Sn (s) + Cd2+(aq)
Q: Consider the reaction, and the following standard reduction potentials: Zn +2 Zn(s) + Cut2 +2 Cu Zn…
A: " Since you have posted multiple questions, we will provide the solution only to the first question…
Q: 19. Based on the information given below, give the group, period, block, and identity of each…
A: The objective of the question is to identify the element.
Q: 2. 'Give the structure of the major product of the following reaction CH3 NaOCH 3 CH₂OH
A: The objective of this question is to show the mechanism of given reaction. The molecule react with…
Q: When two different liquids are mixed or when a gas or solid is dissolved in a liquid, bonds are…
A: The objective of this question is to show the mixing different liquids or dissolving a gas/solid in…
Q: Can you please provide a detailed curved arrow reaction mechanism for this? Please provide a…
A: A question based on reaction mechanisms. For the given reaction the most appropriate reaction…
Q: 1. An iron pellet 1 cm in diameter is subjected to a heat treatment in a vacuum oven at 300 C to…
A: The aim of this question is to explain the time required for the average hydrogen concentration to…
Q: Identify the molecules or ions below as aromatic, non-aromatic, or anti-aromatic.
A: Aromatic, non-aromatic, and anti-aromatic compounds are classifications in organic chemistry based…
Q: Check my work A chemical company wishes to determine whether a new catalyst XA-100 changes the mean…
A: The objective of this question is to determine the effect of a new catalyst, XA , on the hourly…
Step by step
Solved in 3 steps with 1 images

- Using the Frost Circle method to outline the molecular orbitals of cyclobutadiene, and identify whether it is aromatic, antiaromatic or non-aromatic. Explain.Please help! Part 3B Set 1. Can IR spectroscopy be used to differentiate between the two compounds? Briefly explain why or why not. What significant absorptions would be observed in the IR spectrum?How many kinds of chemically non-equivalent hydrogens are there in each of the following compounds?
- Predict the electronic configurations of (a) the benzeneanion, (b) the benzene cation. Estimate the π-bond energy in each case.If the imaginary replacement of either of two protons forms enantiomers, then those protons are said to be enantiotopic. The NMR is not a chiral probe, and it cannot distinguish between enantiotopic protons. They are seen to be “equivalent by NMR.” What other protons in cyclobutanol are enantiotopic?For each of the following reactions, check in the box which of these A or B components represents "highest occupied molecular orbital" (HOMO) and "lowest unoccupied molecular orbital" (LUMO). Once assigned, determine their type of orbitals.
- Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. Long and detailed explanations are not needed. Keep it short, brief, and direct because I only need the answers as soon as possible. Assign the absolute configuration on each of the chiral centers in the compound below.Predict whether the cyclic system is aromatic , anti-aromatic, or non-aromatic by counting electrons and explain why ? Also circle all the basic nitrogen’s wherever possible . Show all lone pairs which are involved in cyclic delocalization?For conjugated C=C with carbonyl group or another double bond, lower frequency for vibration and lower force constant is needed? Why?
- Represent a diastereoisomer of theproposed molecule, indicate theabsolute settings of your carbonschirals and discuss whether this is optically activeor not - justify:Part 3B Set 1. Can 1H NMR spectroscopy be used to differentiate between the two compounds? Briefly explain why or why not. Predict the 1H NMR spectrum for each compound (include integration, multiplicity, and approximate chemical shift). Put it in data table format.which molecule is more symmetrical ? how they distinguish in IR with fact ( without looking at their IR spectrum ?) 1,2-dibromobenzene and 1,3-di bromobenzene

