A carbon sample contains 4000 atoms of radioactive element 235U. Given that 235U has a half-life of 800 years, how long would it take to decay to 150 atoms. Note: Half-life (Ln2) / k O a. -3500 years. O b. 3500 years O c. 4000 years O d. -4000 years Oe. None
A carbon sample contains 4000 atoms of radioactive element 235U. Given that 235U has a half-life of 800 years, how long would it take to decay to 150 atoms. Note: Half-life (Ln2) / k O a. -3500 years. O b. 3500 years O c. 4000 years O d. -4000 years Oe. None
Chapter6: Exponential And Logarithmic Functions
Section6.7: Exponential And Logarithmic Models
Problem 1SE: With what kind of exponential model would half-life be associated? What role does half-life play in...
Related questions
Question
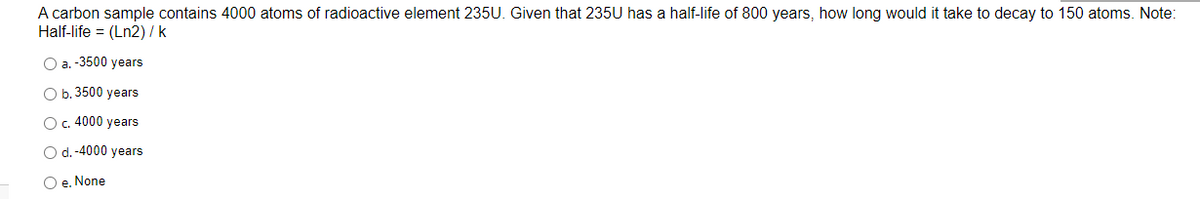
Transcribed Image Text:A carbon sample contains 4000 atoms of radioactive element 235U. Given that 235U has a half-life of 800 years, how long would it take to decay to 150 atoms. Note:
Half-life = (Ln2) / k
O a. -3500 years
O b. 3500 years
O c. 4000 years
O d. -4000 years
O e. None
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you


College Algebra (MindTap Course List)
Algebra
ISBN:
9781305652231
Author:
R. David Gustafson, Jeff Hughes
Publisher:
Cengage Learning


College Algebra (MindTap Course List)
Algebra
ISBN:
9781305652231
Author:
R. David Gustafson, Jeff Hughes
Publisher:
Cengage Learning