(a) Determine the formal charge of oxygen in the following structure. If the atom is formally neutral, indicate a charge of zero. (b) Draw an alternative Lewis (resonance) structure for the compound given in part (a). Show the unshared pairs and nonzero formal charges in your structure. Don't use radicals. Formal charge on O 0
(a) Determine the formal charge of oxygen in the following structure. If the atom is formally neutral, indicate a charge of zero. (b) Draw an alternative Lewis (resonance) structure for the compound given in part (a). Show the unshared pairs and nonzero formal charges in your structure. Don't use radicals. Formal charge on O 0
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter14: Acids And Bases
Section14.8: Acid-base Reactions Of Salts
Problem 14.23CE
Related questions
Question
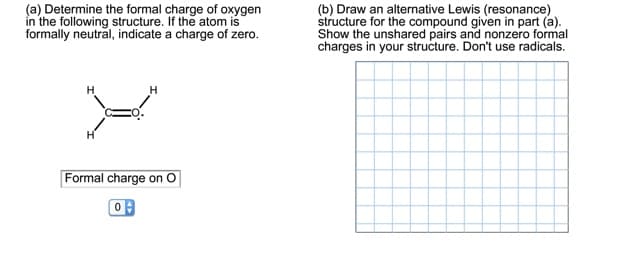
Transcribed Image Text:(a) Determine the formal charge of oxygen
in the following structure. If the atom is
formally neutral, indicate a charge of zero.
(b) Draw an alternative Lewis (resonance)
structure for the compound given in part (a).
Show the unshared pairs and nonzero formal
charges in your structure. Don't use radicals.
Formal charge on O
0
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning