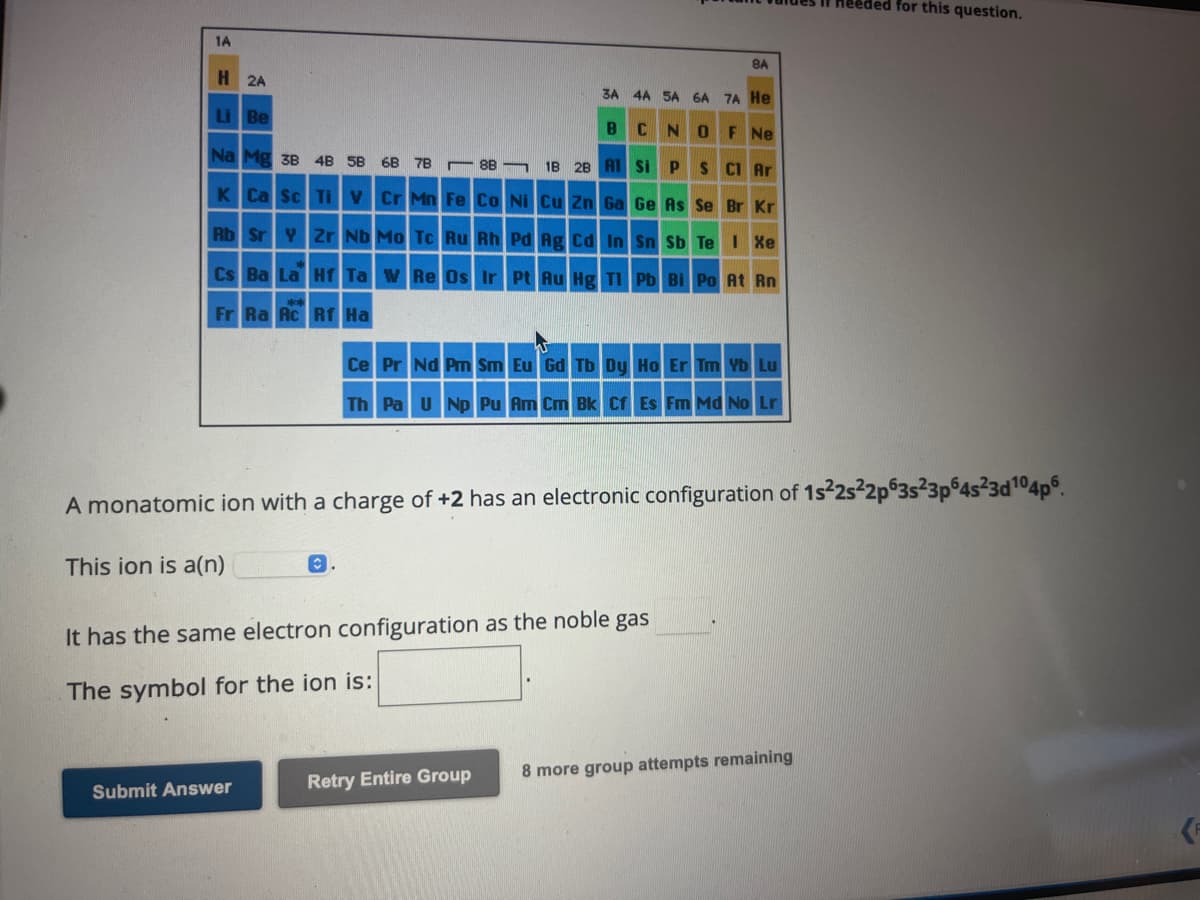
A monatomic ion with a charge of +2 has an electronic configuration of 1s²2s²2p63s²3p64s²3d¹04p6. This ion is a(n) O. It has the same electron configuration as the noble gas The symbol for the ion is:
A monatomic ion with a charge of +2 has an electronic configuration of 1s²2s²2p63s²3p64s²3d¹04p6. This ion is a(n) O. It has the same electron configuration as the noble gas The symbol for the ion is:
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter1: Chemical Foundations
Section: Chapter Questions
Problem 1RQ: Define and e xplain the differences between the following terms. a. law and theory b. theory and...
Related questions
Question
Transcribed Image Text:1A
H2A
Li Be
4A 5A 6A 7A He
CNOF Ne
Cl Ar
Na Mg 3B 48 58 68 7888
Si P S
K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr
Pd Ag Cd In Sn Sb Te I Xe
Rb Sr Y Zr Nb Mo Tc Ru Rh
Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn
Fr Ra Rc Rf Ha
C
Submit Answer
3A
1B 2B Al
8A
Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu
Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr
A monatomic ion with a charge of +2 has an electronic configuration of 1s²2s²2p63s23p64s²3d¹04p6.
This ion is a(n)
It has the same electron configuration as the noble gas
The symbol for the ion is:
Retry Entire Group
eded for this question.
8 more group attempts remaining
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning