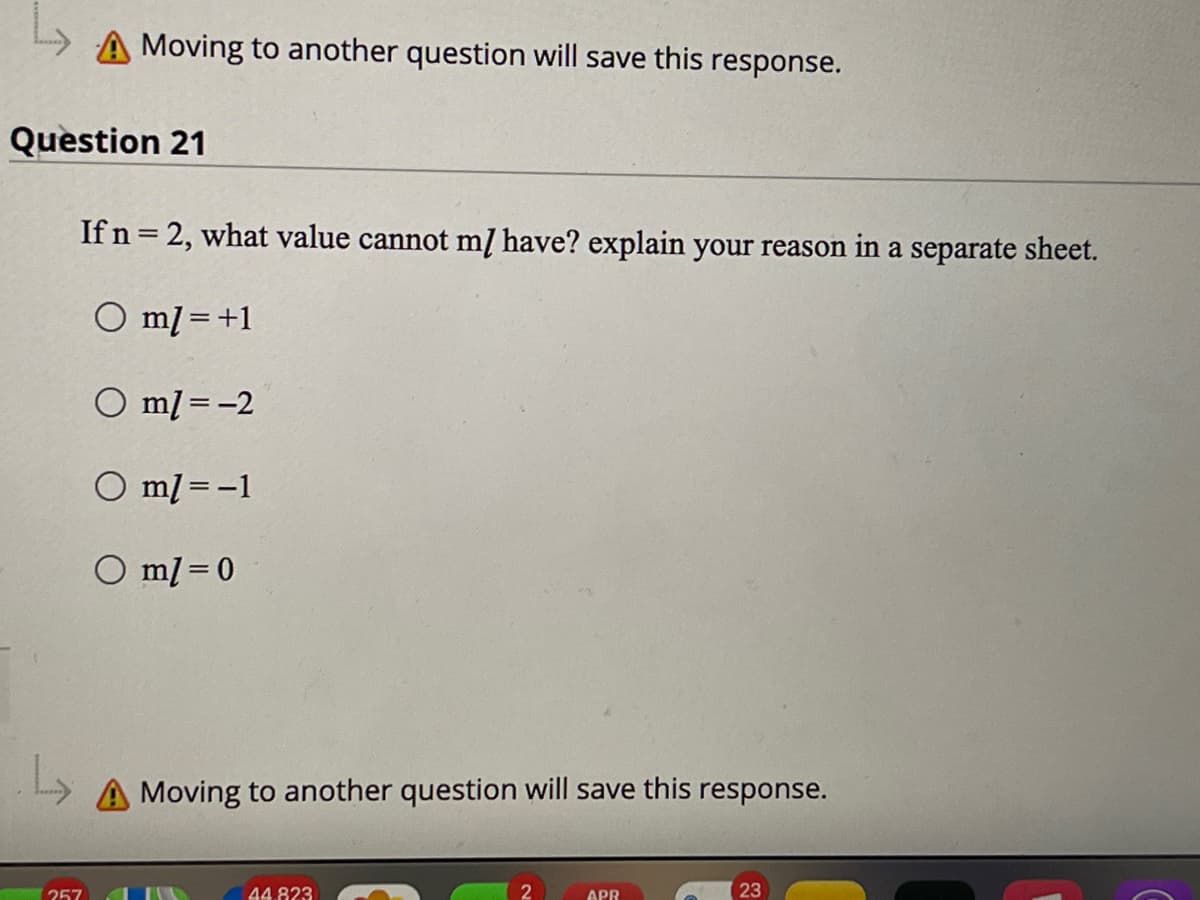
A Moving to another question will save this response. Question 21 If n = 2, what value cannot mĮ have? explain your reason in a separate sheet. O ml =+1 %3D O ml = -2 O ml = -1 O ml = 0 A Moving to another question will save this response. 257 44.823 APR 23
Q: A diprotic acid, H, A, has acid dissociation constants of K 2.32 x 10 and K2 = 5.84 x 10-12.…
A: Hii there, As there are multiple question posted. we are answering first question. If you need…
Q: Consider the following reaction at 100.0 °C: Al(s) + NaOH(aq) + H20(l) ⇄ Na[Al(OH)4](aq) + H2(g) A)…
A: Le Chatelier's principle: This principle is used to predict the effect of a change in conditions on…
Q: Consider the voltaic cell illustrated in the figure (Figure 1), which is based on the cell reaction…
A: G = -nFE = J/mol Zn(s)-->Zn^2+(aq)+2e^- 0.765 V Cu^2+(aq)+2e^- -->Cu(s) 0.337 V n = 2, F =…
Q: Which of the following statements is/are TRUE about the Calorimetry experiment? I. The…
A: following statements are TRUE about the Calorimetry experiment - I. The reactions were kept…
Q: Consider the following reaction: N2(g) + 3H2(g) ⇄ 2NH3(g) A. When would this reaction be…
A: Reaction : N2(g) + 3H2(g) ⇄ 2NH3(g)…
Q: Bismuth-214 undergoes a radioactive decay chain before it reaches nudear stability, forming the inal…
A:
Q: The equilibrium constant, K, for the following reaction is 4.48x10-3 at 676 K. NH4I(s)ENH3(g) +…
A: Kc is the ratio of the equilibrium concentrations of product over equilibrium concentrations of…
Q: Q 1(c) Show the mechanism for the nucleophilic substitution reaction that occurs when 2-bromopropane…
A: Given :- Organic reactions of nucleophillic substitution reaction and nucleophillic addition…
Q: What type of intermolecular force causes the dissolution of RbBr in water? O dispersion forces O…
A:
Q: 5 p.m. on Saturday. And, they are considering offering seniors a 10% discount on all private pay…
A:
Q: Deshielding of the methyl groups attached to a carbonyl causes it to be observed at A) ~10 ppm d.…
A: The proton NMR helps us to get the idea of different types of hydrogens present in an organic…
Q: A voltaic cell employs the following redox reaction: 2 Fe+ (aq) + 3 Mg (s) → 2 Fe (s)+3 Mg²+ (aq)…
A: Given- [Fe+3]=3.10M [Mg2+]=1.2×10-3M Ecell=? ->Answer may differ slightly because values of…
Q: NH2 NH2 NH2 a.
A:
Q: Arrange the following according to increasing boiling point.
A:
Q: wo salts A and B are dissolved in water. At the temperature of the experiment, the solubility of A…
A:
Q: Wine goes bad soon after opening because the ethanol (CH,CH,OH) in it reacts with oxygen gas (0,…
A:
Q: How many moles of O2 gas are produced at 1 atm pressure and 25 °C by the electrolysis of an aqueous…
A:
Q: che single best answer. s pair of elements (Ag and Cd), predict which one has more stable isoto Ag…
A: Protons are positively charged particles and neutrons are particles having no charge and these…
Q: What is the % concentration (m/v) of a solution prepared by dissolving 28.3 of NaOH in enough water…
A: Given, mass of NaOH = 28.3 g Volume of solution = 789 ml % (m/v) = ?
Q: An analytical chemist is titrating 94.4 mL of a 0.5900M solution of ammonia (NH, with a 0.1400M…
A: Buffer solution: A buffer solutions is an aqueous solution consisting of a mixture of a weak base…
Q: Set up a cell that has a Mg electrode in a 1.0 M aqueous Mg?+ solution on the left side and a Cu…
A: Left side of cell reaction represents oxidation Half cell i.e - - > oxidation of Mg Mg is…
Q: 1. CH2=CHLI 2. НзО*
A:
Q: Which of the following statements is true, given the picture provided. a.Yellow dye is more polar…
A: The given picture is an representative example of the thin layer chromatograpy with various coloured…
Q: Calculate the molar concentration of Ba2+ in saturated BaC2O4 solution. The pH was kept at 4.5. Ksp…
A: Given: pH = 4.5 Ksp of BaC2O4 = 1.0x10-6 Ka1 of H2C2O4 = 5.4x10-2 Ka2 of H2C2O4 = 5.42x10-5
Q: Arrange the oxoacids of chlorine according to strength. Strongest acid Weakest acid Answer Bank…
A: The acidity order of Oxo acids are given below
Q: Part A The Ksp of Zn(OH)2 is 1.8x10-14. Find Ecell for the following half-reaction. Zn(OH)2(s) +…
A: This can be solved as follows
Q: 16 of 21 Review Constants | Periodic Table Part B Calculate the pH of a 0.10 M solution of…
A:
Q: Calculate the pH of a 0.020 M solution of phenylacetic acid, C6H5CH2COOH. What will be the pH if the…
A:
Q: What is the molar mass of an unknown gas with a density of 2.00 g/L at 1.00 atm and 25.0 °C?
A:
Q: A solution is prepared at 25 °C that is initially 0.26M in methylamine (CH,NH,), a weak base with…
A: this is a basic buffer , so we will use the formula of basic buffer.
Q: Draw in the missing structures A and B, and then choose what could function as reagent C to complete…
A: In this question, we have to draw the missing structure A,B and the reagent C.
Q: Part A How much concentrated solution would it take to prepare 2.95 L of 0.585 M HCl upon dilution…
A:
Q: In the context of chemistry and rate laws. What assumptions do we make when analyzing rate law…
A: The steady-state approximation is a method used to derive a rate law. The steady state…
Q: Provide an efficient synthesis of each molecule using the starting materials indicated. While no…
A: Here we have to synthesize the following given compounds from the starting material through mult
Q: Ag2CO3 (s) 2Ag+(aq) + CO3-2 (aq) G= -64.88J/mol b. CaF2(s) Ca+2(aq) + 2F-(aq) G= -59.32J/mol…
A: From the reaction between equilibrium constant and Gibbs free energy change is □G = - RT × ln(K)…
Q: This is one question in two parts. Same reactant in both, but change in reagent. Draw the major…
A:
Q: 0.250 mol of N2 is placed into a 0.750 L container. What is the density of the gas? 84.1 g/L 4.67…
A:
Q: A colleague produces this graph of the concentration of carbon dioxide as a function of time. In the…
A:
Q: 1. Give the major product(s) of the following reactions as needed. OH 1. PCC ОН 2. НО Jones ОН HO…
A:
Q: Calculate entropy change 1. combustion of one gram of methane to form carbon dioxide and liquid…
A:
Q: 7) Draw both the condensed and line bond structures for A) 1,3,3-trimethyl cyclohexene B)…
A:
Q: (i). Using the reaction Zn(s) + 2Co3+ (aq) -› Zn?+ (aq) + 2Co2+ (aq) as an example, draw a diagram…
A: The electrochemical cell reaction is given as, Zn(s)+2Co3+(aq)→Zn2+(aq)+2Co2+(aq) From electro…
Q: Consider the following reaction between iodate and iodide ions. 10, (aq) + 5 I (aq) + 6 H* (aq) → 3…
A: The rate of a reaction can be increased by, • Adding a catalyst - A catalyst decreases the…
Q: What is the phenomenon that occurs when excited gaseous elements emit only a few colored lines? O…
A:
Q: . If 45.0g of H20 react, how many moles, grams and molecules of H3PO. are produced PCIS H2O H&PO.…
A: Recall the reaction, PCl5 + H2O -----> H3PO4 + HCl mass of H2O= 45.0 g molar mass of H2O = 18 g…
Q: Here is the reaction equation for the combustion of butane, which is an exothermic reaction. 2…
A: Exothermic reaction that releases energy from the system to its surroundings
Q: Be sure to answer all parts. Determine the percent ionization of the following solutions of phenol…
A:
Q: The rate constant of Iodine-131 is 8.66 x 10^-2 per day. 1. What is the half-life of I-131? 2. If…
A: As the unit of rate constant is time-1, it is a first order reaction. For a first order reaction,…
Q: Calculate the volume of 1.00M acetic acid and mass of sodium bicarbonate needed to make 50.0ml of a…
A:
Q: Decide whether or not each of the following metals dissolves in 1 M HNO3. For those metals that do…
A: We have to tell what happened when Al is dissolved in 1 M HNO3.
Step by step
Solved in 2 steps with 2 images

- Calculate the average density of a single Al-27 atom by assuming that it is a sphere with a radius of 0.143 nm. The masses of a proton, electron, and neutron are 1.67261024g,9.10941028g, and 1.67491024g, respectively. The volume of a sphere is 4r3/3, where r is its radius. Express the answer in grams per cubic centimeter. The density of aluminum is found experimentally to be 2.70 g/cm3. What does that suggest about the packing of aluminum atoms in the metal?Scenario In an investigation, lab groups combine two compounds, as shown below. Based on the data collected from all lab groups, you realize that one group’s data was not possible. Mg (s) + 2HCl (aq) → MgCl2 (aq) + H2 (g) External Data Group Number Mass of Mg (g) Mass of HCl (g) Mass of MgCl2 (g) 1 5 15 20 2 5 15 17 3 5 15 21 4 5 15 20 5 5 15 19 Prompt Make a claim about which group’s data is not possible. Use evidence and scientific reasoning to support your claim. Include a rebuttal in your answer. Claim (1 sentence): ______________________________________________________________. Evidence 1 (2-3 sentences):…Consider the table. Metal Tm (K) ΔHfus (kJ/mol) Tb (K) ΔHvap (kJ/mol) LiLi 454 2.99 1615 134.7 NaNa 371 2.60 1156 89.6 KK 336 2.33 1033 77.1 RbRb 312 2.34 956 69 CsCs 302 2.10 942 66 Using the data, calculate Δ?fusΔSfus and Δ?vapΔSvap for Cs.Cs. Δ?vap=ΔSvap= JK⋅molJK⋅mol Δ?fus=ΔSfus= JK⋅molJK⋅mol
- Perform the following mathematical function.51.11 + (0.97/2.414 × 0.32150 × e.-3.25) + 1.52444 3.4 The written value of 1.0 μm meansa. 102 mb. 10-2 mc. 106 m d. 10-6 m e. 10-9 m 1.2 An example of a physical property isA) the reaction of rubidium with water to form rubidium hydroxideB) the density of boronC) the burning of sulfur to form sulfur dioxideD) the reaction of cesium with oxygen to form cesium superoxideE) the energy content of liquid sodiumAm I right? Sample 1 2 3 Calculations for sample 1 Mass of aluminum dish 1.24g 1.26g 1.24g - Mass of aluminum dish + Epsom salt 1.70g 1.94g 1.80g - Epsom salt, original (hydrated) mass 0.46g 0.68g 0.56g 1.70g – 1.24g = 0.46g Mass of aluminum dish + Epsom salt after heating 1.42g 1.62g 1.61g Epsom salt, heated (dehydrated) mass 0.18g 0.36g 0.37g 1.42g- 1.24g = 0.18g Mass of water 0.28g 0.32g 0.19g 0.46g – 0.18g = 0.28g Moles of magnesium sulfate 0.0015mol 0.0030mol 0.0031mol 0.18g x (1mol/120.37g)= 0.0015mol Moles of water 0.016mol 0.018mol 0.011mol 0.28g x (1mol/18.02g) = 0.016mol Ratio of moles water: moles of magnesium sulfate 32:3 18:3 11:3.1 0.016 : 0.0015= 32:3 Average Ratio Knowing that magnesium sulfate is heptahydrate, what is the % error on the mass percentage of water? Show all calculations. --> percentage error: 30.43% 1)…2Cr(s) + 3Cu2+(aq) ⟶ 2Cr3+(aq) + 3Cu(s) Calculate the Ecell
- Probably meant in the calculation that 20*10K+20=0.6, but how did you get that to be 20=6K+12?Mass of the gelatin capsule was: 0.1134 g Mass of the capsule and the allow sample was: 0.3218 g Ambient Temperature was: 23°C Mass of empty beaker: 154.3 g Mass of beaker with water: 401.6 g Vapor pressure of water for this data: 21.1 mmHg Atmospheric Pressure: 732 mmHg Assume density of water to be 1.0000 g/mL What is the volume of the displaced water in Liters? Using PH2V= nH2RT, nH2=PH2V/RT how many moles of H2 were evolved? Determine the number of moles of H2 per gram of sample. Determine the %Al using NH2= (%AL/100 * 0.0556) + (100-%AL/100 * 0.0153)During a titration experiment, 50.00 mL of a certain solution needs to be transferred to an Erlenmeyer flask. To measure the volume, a student used a calibrated 10-mL pipette five times. If the pipette delivers an actual volume of 9.98 mL with an uncertainty of ±0.02 mL, what will be the uncertainty in the final volume?
- 21,1) What percentage of 145X (t1/2 = 768.7 h) would remain after 505.4 hours. Use the formula with 0.693 instead of ln 2. The data for element X does not exist. All numbers are hypotheticalA loss of 0.35 mg of Zn occurs in the course of analysis to determine the element. Calculate the percent relative error due to this loss if the masses of Zn in the replicate samples are the ff: (a) 40.0 mg (b) 400.0 mg (c) 175.0 mg (d) 600.0 mg.1.state the process determining the volume of irregular object 2.In determining the density of an irregular stone of a mass 80g.The stone was dip in a measuring cylinder fill with water and gave a find reading of 100cm3 after the experiment. The density of a stone was 2g/cm2.Find the initial reading of the measuring cylinder. 3.A solid of mass 450kg has a volume of 20cm3 calculate the relate density of solid. Density of water:100pkg/m3 changes in cm

