A patient's serum magnesium level is reported by the lab as: 1.6 mMol/L. Convert this value to mEq/L. Round to one decimal place, do not enter unita Cation Formula Valence Formula weight Anion Formula Valence Formula weight Aluminum A13+ 27 Acetate C,H;O2 1 59 Ammonium NH, 1 18 Bicarbonate HCO, 61 Calcium Ca2+ 2 40 Carbonate co,2- 2 60 Ferric Fe3+ 3 56 Chloride 35.5 Ferrous Fe2+ 56 Citrate CH;0,3- 3 189 Lithium Lit 1 7 Gluconate CH1,07 1 195 Magnesium Mg2+ 2 24 Hydroxide OH- 1 17 Potassium K+ 39 Lactate 89 Sodium Na+ 1 23 H,PO, Phosphate, monobasic 1 97 Phosphate dibasic HPO,2- 2 96 Sulfate So,2- 2 96
A patient's serum magnesium level is reported by the lab as: 1.6 mMol/L. Convert this value to mEq/L. Round to one decimal place, do not enter unita Cation Formula Valence Formula weight Anion Formula Valence Formula weight Aluminum A13+ 27 Acetate C,H;O2 1 59 Ammonium NH, 1 18 Bicarbonate HCO, 61 Calcium Ca2+ 2 40 Carbonate co,2- 2 60 Ferric Fe3+ 3 56 Chloride 35.5 Ferrous Fe2+ 56 Citrate CH;0,3- 3 189 Lithium Lit 1 7 Gluconate CH1,07 1 195 Magnesium Mg2+ 2 24 Hydroxide OH- 1 17 Potassium K+ 39 Lactate 89 Sodium Na+ 1 23 H,PO, Phosphate, monobasic 1 97 Phosphate dibasic HPO,2- 2 96 Sulfate So,2- 2 96
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter13: The Chemistry Of Solutes And Solutions
Section: Chapter Questions
Problem 57QRT
Related questions
Question
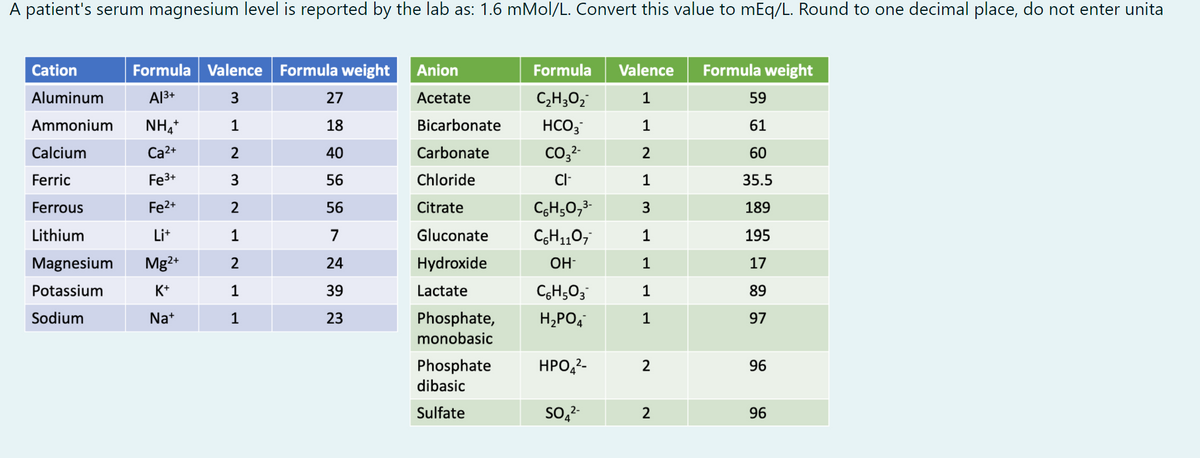
Transcribed Image Text:A patient's serum magnesium level is reported by the lab as: 1.6 mMol/L. Convert this value to mEq/L. Round to one decimal place, do not enter unita
Cation
Formula Valence Formula weight
Anion
Formula
Valence
Formula weight
Aluminum
Al3+
27
Acetate
C,H;O,
1
59
Ammonium
NH,+
1
18
Bicarbonate
HCO;
1
61
Calcium
Ca2+
40
Carbonate
Co32-
2
60
Ferric
Fe3+
3
56
Chloride
Cl-
1
35.5
Ferrous
Fe2+
56
Citrate
CgH;0,3-
3
189
Lithium
Lit
1
7
Gluconate
CH110,
1
195
Magnesium
Mg2+
24
Hydroxide
OH-
1
17
Potassium
K+
1
39
Lactate
CGH503
1
89
Sodium
Na+
1
23
Phosphate,
H,PO,
1
97
monobasic
Phosphate
НРО,2-
2
96
dibasic
Sulfate
So,2-
2
96
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning