A solution of tartaric acid (H2C,H,O6) with a known concentration of 0.155 M H2C,H,O6 is titrated with a 0.425 M NAOH solution. How many mL of NaOH are required to reach the second equivalence point with a starting volume of 70.0 mL H;C,H,O6 , according to the following balanced chemical equation: H2C,H,O6 + 2 NaOH – NazC4H4O6 + 2 H2o
A solution of tartaric acid (H2C,H,O6) with a known concentration of 0.155 M H2C,H,O6 is titrated with a 0.425 M NAOH solution. How many mL of NaOH are required to reach the second equivalence point with a starting volume of 70.0 mL H;C,H,O6 , according to the following balanced chemical equation: H2C,H,O6 + 2 NaOH – NazC4H4O6 + 2 H2o
Chapter16: Applications Of Neutralization Titrations
Section: Chapter Questions
Problem 16.48QAP
Related questions
Question
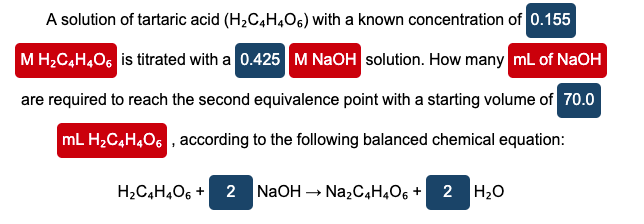
Transcribed Image Text:A solution of tartaric acid (H2C,H,O6) with a known concentration of 0.155
M H;C,H406 is titrated with a 0.425 M NaOH solution. How many mL of NaOH
are required to reach the second equivalence point with a starting volume of 70.0
mL H,C4H,O6 , according to the following balanced chemical equation:
H2C4H,O6 +
NaOH -
Na,C4H,O6 + 2 H20
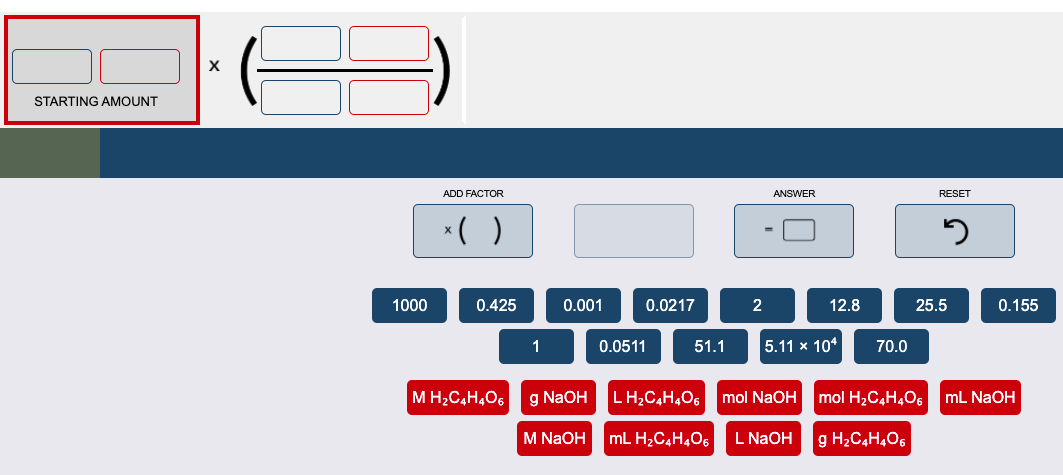
Transcribed Image Text:STARTING AMOUNT
ADD FACTOR
ANSWER
RESET
*( )
1000
0.425
0.001
0.0217
2
12.8
25.5
0.155
1
0.0511
51.1
|5.11 х 104
70.0
M H2C,H,O6
g NaOH
LH,C,H,O6
mol NaOH
mol H2C4H,O6
mL NaOH
M NaOH
mL H2C,H4O6
L NAOH
g H2C,H,O6
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning