A steel tank contains 273 g of ammonia gs (NH3 with molar mass 17.0 g/mol) at a pressure of 1.53 x 106 and a temperature of 53.0°C. (a) What is the volume of the tank in liters? (b) Later the temperature is 60.8°C and the pressure is 1.10 x 10° Pa. How many grams of gas have leaked out of the tank? (Ignore the thermal contraction of the tank.) (a) Number Units (b) Number i Units
A steel tank contains 273 g of ammonia gs (NH3 with molar mass 17.0 g/mol) at a pressure of 1.53 x 106 and a temperature of 53.0°C. (a) What is the volume of the tank in liters? (b) Later the temperature is 60.8°C and the pressure is 1.10 x 10° Pa. How many grams of gas have leaked out of the tank? (Ignore the thermal contraction of the tank.) (a) Number Units (b) Number i Units
Chapter2: The Kinetic Theory Of Gases
Section: Chapter Questions
Problem 22P: A cubic container of volume 2.00 L holds 0.500 mol of nitrogen gas at a temperature of 25.0 . What...
Related questions
Question
100%
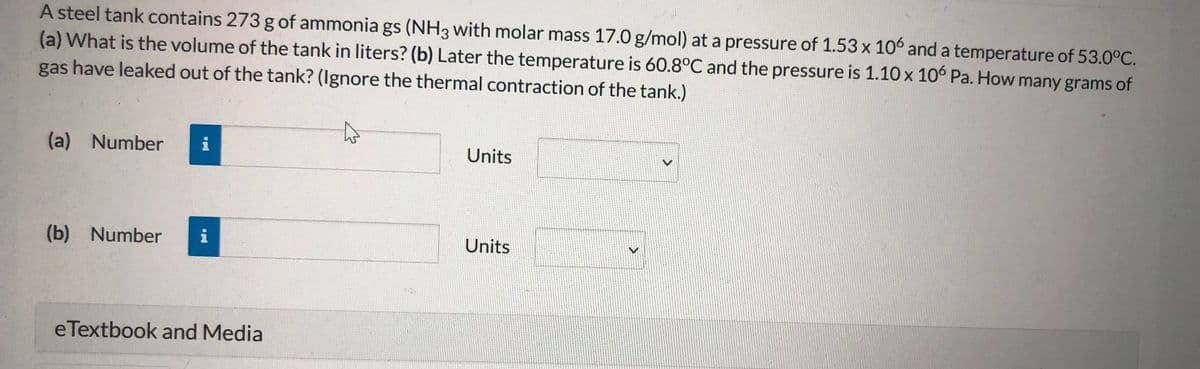
Transcribed Image Text:A steel tank contains 273 g of ammonia gs (NH3 with molar mass 17.0 g/mol) at a pressure of 1.53 x 106 and a temperature of 53.0°C.
(a) What is the volume of the tank in liters? (b) Later the temperature is 60.8°C and the pressure is 1.10 x 10° Pa. How many grams of
gas have leaked out of the tank? (Ignore the thermal contraction of the tank.)
(a) Number
i
Units
(b) Number
Units
eTextbook and Media
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning


Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning